Systemic Inflammation Causes Microglial Dysfunction With a Vascular AD phenotype
- PMID: 36704658
- PMCID: PMC9871075
- DOI: 10.1016/j.bbih.2022.100568
Systemic Inflammation Causes Microglial Dysfunction With a Vascular AD phenotype
Abstract
Background: Studies in rodents and humans have indicated that inflammation outside CNS (systemic inflammation) affects brain homeostasis contributing to neurodevelopmental disorders. Itis becoming increasingly evident that such early insults may also belinked to neurodegenerative diseases like late-onset Alzheimer's disease (AD). Importantly, lifestyle and stress, such as viral or bacterial infection causing chronic inflammation, may contribute to neurodegenerative dementia. Systemic inflammatory response triggers a cascade of neuroinflammatory responses, altering brain transcriptome, cell death characteristic of AD, and vascular dementia. Our study aimed to assess the temporal evolution of the pathological impact of systemic inflammation evoked by prenatal and early postnatal peripheral exposure of viral mimetic Polyinosinic:polycytidylic acid (PolyI:C) and compare the hippocampal transcriptomic changes with the profiles of human post-mortem AD and vascular dementia brain specimens.
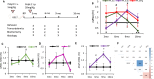
Methods: We have engineered the PolyI:C sterile infection model in wildtype C57BL6 mice to achieve chronic low-grade systemic inflammation. We have conducted a cross-sectional analysis of aging PolyI:C and Saline control mice (3 months, 6 months, 9 months, and 16 months), taking the hippocampus as a reference brain region, and compared the brain aging phenotype to AD progression in humans with mild AD, severe AD, and Controls (CTL), in parallel to Vascular dementia (VaD) patients' specimens.
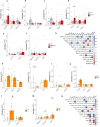
Results: We found that PolyI:C mice display both peripheral and central inflammation with a peak at 6 months, associated with memory deficits. The hippocampus is characterized by a pronounced and progressive tauopathy. In PolyI:C brains, microglia undergo aging-dependent morphological shifts progressively adopting a phagocytic phenotype. Transcriptomic analysis reveals a profound change in gene expression throughout aging, with a peak in differential expression at 9 months. We show that the proinflammatory marker Lcn2 is one of the genes with the strongest upregulation in PolyI:C mice upon aging. Validation in brains from patients with increasing severity of AD and VaD shows the reproducibility of some gene targets in vascular dementia specimens as compared to AD ones.
Conclusions: The PolyI:C model of sterile infection demonstrates that peripheral chronic inflammation causes progressive tau hyperphosphorylation, changes in microglia morphology, astrogliosis and gene reprogramming reflecting increased neuroinflammation, vascular remodeling, and the loss of neuronal functionality seen to some extent in human AD and Vascular dementia suggesting early immune insults could be crucial in neurodegenerative diseases.
Keywords: Brain aging; Inflammation; Microglia; PolyI:C; Sporadic Alzheimer's disease; Vascular dementia.
© 2022 Published by Elsevier Inc.
Conflict of interest statement
There are no competing interests.
Figures





References
LinkOut - more resources
Full Text Sources
Miscellaneous

