Extracellular histone H3 facilitates ferroptosis in sepsis through ROS/JNK pathway
- PMID: 36705411
- PMCID: PMC9795329
- DOI: 10.1002/iid3.754
Extracellular histone H3 facilitates ferroptosis in sepsis through ROS/JNK pathway
Abstract
Introduction: Previous evidence realized the critical role of histone in disease control. The anti-inflammatory function of estradiol (E2) in sepsis has been documented. We here intended to unveil the role of extracellular histone H3 in sepsis regarding cell ferroptosis and the role of E2 in a such mechanism.
Methods: Clinical sample, cecal ligation and puncture (CLP)-induced animal models and lipopolysaccharides (LPS)-induced cell models were prepared for testing relative expression of extracellular histone H3 and E2 as well as analyzing the role of extracellular histone H3 and E2 in sepsis concerning cell viability, reactive oxygen species (ROS), and ferroptosis.
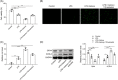
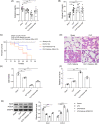
Results: Under sepsis, we found increased ferroptosis and extracellular histone H3 content, but reduced E2 concentration. Extracellular histone H3 facilitated ferroptosis of human umbilical vein endothelial cells (HUVECs) induced by LPS through activating the ROS/c-Jun N-terminal kinase (JNK) pathway. Moreover, E2 antagonized the effect of extracellular histone H3 on LPS-induced HUVEC ferroptosis and sepsis injury in CLP-induced animal models.
Conclusion: We highlighted that extracellular histone H3 facilitated lipopolysaccharides-induced HUVEC ferroptosis via activating ROS/JNK pathway, and such an effect could be antagonized by E2.
Keywords: c-Jun N-terminal kinase; estradiol; ferroptosis; histones H3; reactive oxygen species; sepsis.
© 2022 The Authors. Immunity, Inflammation and Disease published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
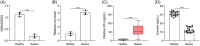


Similar articles
-
Septic serum mediates inflammatory injury in human umbilical vein endothelial cells via reactive oxygen species, mitogen activated protein kinases and nuclear factor‑κB.Int J Mol Med. 2021 Jan;47(1):267-275. doi: 10.3892/ijmm.2020.4785. Epub 2020 Nov 10. Int J Mol Med. 2021. PMID: 33236149 Free PMC article.
-
NOD2-mediated HDAC6/NF-κb signalling pathway regulates ferroptosis induced by extracellular histone H3 in acute liver failure.J Cell Mol Med. 2022 Nov;26(21):5528-5538. doi: 10.1111/jcmm.17582. Epub 2022 Oct 12. J Cell Mol Med. 2022. PMID: 36226351 Free PMC article.
-
Irisin protects against sepsis-associated encephalopathy by suppressing ferroptosis via activation of the Nrf2/GPX4 signal axis.Free Radic Biol Med. 2022 Jul;187:171-184. doi: 10.1016/j.freeradbiomed.2022.05.023. Epub 2022 Jun 1. Free Radic Biol Med. 2022. PMID: 35660523
-
[Role of Nrf2/GPX4 mediated ferroptosis in intestinal injury in sepsis].Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2023 Nov;35(11):1188-1194. doi: 10.3760/cma.j.cn121430-20230616-00451. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2023. PMID: 37987130 Chinese.
-
Inhibition of STEAP1 ameliorates inflammation and ferroptosis of acute lung injury caused by sepsis in LPS-induced human pulmonary microvascular endothelial cells.Mol Biol Rep. 2023 Jul;50(7):5667-5674. doi: 10.1007/s11033-023-08403-7. Epub 2023 May 20. Mol Biol Rep. 2023. PMID: 37209327
Cited by
-
Isoelectric trapping and discrimination of histones from plasma in a microfluidic device using dehydrated isoelectric gate.Mikrochim Acta. 2024 Feb 13;191(3):131. doi: 10.1007/s00604-024-06223-5. Mikrochim Acta. 2024. PMID: 38351209
References
-
- Purcarea A, Sovaila S. Sepsis, a 2020 review for the internist. Rom J Intern Med. 2020;58(3):129‐137. - PubMed
-
- Jacobi J. The pathophysiology of sepsis—2021 update: part 2, organ dysfunction and assessment. Am J Health Syst Pharm. 2022;79(6):424‐436. - PubMed
-
- Hunt A. Sepsis: an overview of the signs, symptoms, diagnosis, treatment and pathophysiology. Emerg Nurse. 2019;27(5):32‐41. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

