p53 Inhibition in Pancreatic Progenitors Enhances the Differentiation of Human Pluripotent Stem Cells into Pancreatic β-Cells
- PMID: 36707464
- PMCID: PMC10185607
- DOI: 10.1007/s12015-023-10509-1
p53 Inhibition in Pancreatic Progenitors Enhances the Differentiation of Human Pluripotent Stem Cells into Pancreatic β-Cells
Abstract
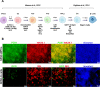
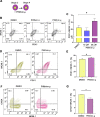
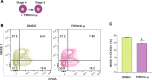
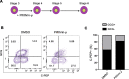
The multipotent pancreatic progenitor cells (MPCs) co-expressing the transcription factors, PDX1 and NKX6.1, are the source of functional pancreatic β-cells. The aim of this study was to examine the effect of p53 inhibition in MPCs on the generation of PDX1+/NKX6.1+ MPCs and pancreatic β-cell generation. Human embryonic stem cells (hESCs) were differentiated into MPCs and β-cells. hESC-MPCs (stage 4) were treated with different concentrations of p53 inhibitors, and their effect was evaluated using different approaches. NKX6.1 was overexpressed during MPCs specification. Inhibition of p53 using pifithrin-μ (PFT-μ) at the MPC stage resulted in a significant increase in the number of PDX1+/NKX6.1+ cells and a reduction in the number of CHGA+/NKX6.1- cells. Further differentiation of MPCs treated with PFT-μ into pancreatic β-cells showed that PFT-μ treatment did not significantly change the number of C-Peptide+ cells; however, the number of C-PEP+ cells co-expressing glucagon (polyhormonal) was significantly reduced in the PFT-μ treated cells. Interestingly, overexpression of NKX6.1 in hESC-MPCs enhanced the expression of key MPC genes and dramatically suppressed p53 expression. Our findings demonstrated that the p53 inhibition during stage 4 of differentiation enhanced MPC generation, prevented premature endocrine induction and favored the differentiation into monohormonal β-cells. These findings suggest that adding a p53 inhibitor to the differentiation media can significantly enhance the generation of monohormonal β-cells.
Keywords: Differentiation protocol; Insulin; Islets; Monohormonal β-cells; NKX6.1; hESCs.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests in this manuscript.
Figures

Similar articles
-
Differentiation of human pluripotent stem cells into two distinct NKX6.1 populations of pancreatic progenitors.Stem Cell Res Ther. 2018 Apr 3;9(1):83. doi: 10.1186/s13287-018-0834-0. Stem Cell Res Ther. 2018. PMID: 29615106 Free PMC article.
-
NKX6.1 transcription factor: a crucial regulator of pancreatic β cell development, identity, and proliferation.Stem Cell Res Ther. 2020 Oct 29;11(1):459. doi: 10.1186/s13287-020-01977-0. Stem Cell Res Ther. 2020. PMID: 33121533 Free PMC article. Review.
-
PDX1- /NKX6.1+ progenitors derived from human pluripotent stem cells as a novel source of insulin-secreting cells.Diabetes Metab Res Rev. 2021 Jul;37(5):e3400. doi: 10.1002/dmrr.3400. Epub 2020 Sep 2. Diabetes Metab Res Rev. 2021. PMID: 32857429
-
Enhanced differentiation of human pluripotent stem cells into pancreatic progenitors co-expressing PDX1 and NKX6.1.Stem Cell Res Ther. 2018 Jan 23;9(1):15. doi: 10.1186/s13287-017-0759-z. Stem Cell Res Ther. 2018. PMID: 29361979 Free PMC article.
-
PDX1, Neurogenin-3, and MAFA: critical transcription regulators for beta cell development and regeneration.Stem Cell Res Ther. 2017 Nov 2;8(1):240. doi: 10.1186/s13287-017-0694-z. Stem Cell Res Ther. 2017. PMID: 29096722 Free PMC article. Review.
Cited by
-
Advancements and challenges in stem cell transplantation for regenerative medicine.Heliyon. 2024 Aug 10;10(16):e35836. doi: 10.1016/j.heliyon.2024.e35836. eCollection 2024 Aug 30. Heliyon. 2024. PMID: 39247380 Free PMC article. Review.
References
-
- Ryan EA, Lakey JR, Rajotte RV, Korbutt GS, Kin T, Imes S, Rabinovitch A, Elliott JF, Bigam D, Kneteman NM, Warnock GL, Larsen I, Shapiro AM. Clinical outcomes and insulin secretion after islet transplantation with the Edmonton protocol. Diabetes. 2001;50:710–719. doi: 10.2337/diabetes.50.4.710. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

