A first-in-class HBO1 inhibitor WM-3835 inhibits castration-resistant prostate cancer cell growth in vitro and in vivo
- PMID: 36709328
- PMCID: PMC9884225
- DOI: 10.1038/s41419-023-05606-5
A first-in-class HBO1 inhibitor WM-3835 inhibits castration-resistant prostate cancer cell growth in vitro and in vivo
Abstract
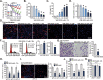
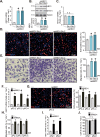
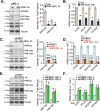
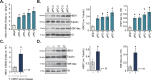
The prognosis and overall survival of castration-resistant prostate cancer (CRPC) patients are poor. The search for novel and efficient anti-CRPC agents is therefore extremely important. WM-3835 is a cell-permeable, potent and first-in-class HBO1 (KAT7 or MYST2) inhibitor. Here in primary human prostate cancer cells-derived from CRPC patients, WM-3835 potently inhibited cell viability, proliferation, cell cycle progression and in vitro cell migration. The HBO1 inhibitor provoked apoptosis in the prostate cancer cells. It failed to induce significant cytotoxicity and apoptosis in primary human prostate epithelial cells. shRNA-induced silencing of HBO1 resulted in robust anti-prostate cancer cell activity as well, and adding WM-3835 failed to induce further cytotoxicity in the primary prostate cancer cells. Conversely, ectopic overexpression of HBO1 further augmented primary prostate cancer cell proliferation and migration. WM-3835 inhibited H3-H4 acetylation and downregulated several pro-cancerous genes (CCR2, MYLK, VEGFR2, and OCIAD2) in primary CRPC cells. Importantly, HBO1 mRNA and protein levels are significantly elevated in CRPC tissues and cells. In vivo, daily intraperitoneal injection of WM-3835 potently inhibited pPC-1 xenograft growth in nude mice, and no apparent toxicities detected. Moreover, intratumoral injection of HBO1 shRNA adeno-associated virus (AAV) suppressed the growth of primary prostate cancer xenografts in nude mice. H3-H4 histone acetylation and HBO1-dependent genes (CCR2, MYLK, VEGFR2, and OCIAD2) were remarkably decreased in WM-3835-treated or HBO1-silenced xenograft tissues. Together, targeting HBO1 by WM-3835 robustly inhibits CRPC cell growth.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
The histone acetyltransferase HBO1 functions as a novel oncogenic gene in osteosarcoma.Theranostics. 2021 Mar 4;11(10):4599-4615. doi: 10.7150/thno.55655. eCollection 2021. Theranostics. 2021. PMID: 33754016 Free PMC article.
-
HBO1 induces histone acetylation and is important for non-small cell lung cancer cell growth.Int J Biol Sci. 2022 May 9;18(8):3313-3323. doi: 10.7150/ijbs.72526. eCollection 2022. Int J Biol Sci. 2022. PMID: 35637972 Free PMC article.
-
PDLIM2 suppression efficiently reduces tumor growth and invasiveness of human castration-resistant prostate cancer-like cells.Prostate. 2016 Feb 15;76(3):273-85. doi: 10.1002/pros.23118. Epub 2015 Oct 26. Prostate. 2016. PMID: 26499308
-
HBO1 overexpression is important for hepatocellular carcinoma cell growth.Cell Death Dis. 2021 May 26;12(6):549. doi: 10.1038/s41419-021-03818-1. Cell Death Dis. 2021. PMID: 34039960 Free PMC article.
-
Multifunctional acyltransferase HBO1: a key regulatory factor for cellular functions.Cell Mol Biol Lett. 2024 Nov 14;29(1):141. doi: 10.1186/s11658-024-00661-y. Cell Mol Biol Lett. 2024. PMID: 39543485 Free PMC article. Review.
Cited by
-
Identification of prognostic hub genes and functional role of BAIAP2L2 in prostate cancer progression: a transcriptomic and experimental study.Front Immunol. 2025 Apr 28;16:1543476. doi: 10.3389/fimmu.2025.1543476. eCollection 2025. Front Immunol. 2025. PMID: 40356901 Free PMC article.
-
MYST2 histone acetyltransferase promotes lung adenocarcinoma progression by regulating the p38 MAPK signaling pathway.Transl Oncol. 2025 Jan;51:102218. doi: 10.1016/j.tranon.2024.102218. Epub 2024 Nov 27. Transl Oncol. 2025. PMID: 39603207 Free PMC article.
-
Targeting SphK1/2 by SKI-178 inhibits prostate cancer cell growth.Cell Death Dis. 2023 Aug 21;14(8):537. doi: 10.1038/s41419-023-06023-4. Cell Death Dis. 2023. PMID: 37604912 Free PMC article.
-
Competitive antagonism of KAT7 crotonylation against acetylation affects procentriole formation and colorectal tumorigenesis.Nat Commun. 2025 Mar 10;16(1):2379. doi: 10.1038/s41467-025-57546-7. Nat Commun. 2025. PMID: 40064919 Free PMC article.
-
Gαi3: a crucial biomarker and therapeutic target in bladder cancer.NPJ Precis Oncol. 2025 Jun 13;9(1):181. doi: 10.1038/s41698-025-00989-4. NPJ Precis Oncol. 2025. PMID: 40514424 Free PMC article.
References
-
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA Cancer J Clin. 2021;71:7–33. - PubMed
-
- Sandhu S, Moore CM, Chiong E, Beltran H, Bristow RG, Williams SG. Prostate cancer. Lancet. 2021;398:1075–90. - PubMed
-
- Attard G, Parker C, Eeles RA, Schroder F, Tomlins SA, Tannock I, et al. Prostate cancer. Lancet. 2016;387:70–82. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

