Mechanical loading is required for initiation of extracellular matrix deposition at the developing murine myotendinous junction
- PMID: 36709857
- PMCID: PMC10218368
- DOI: 10.1016/j.matbio.2023.01.003
Mechanical loading is required for initiation of extracellular matrix deposition at the developing murine myotendinous junction
Abstract
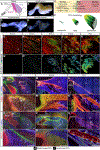
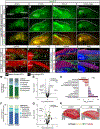
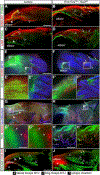
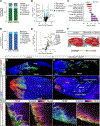
The myotendinous junction (MTJ) contributes to the generation of motion by connecting muscle to tendon. At the adult MTJ, a specialized extracellular matrix (ECM) is thought to contribute to the mechanical integrity of the muscle-tendon interface, but the factors that influence MTJ formation during mammalian development are unclear. Here, we combined 3D imaging and proteomics with murine models in which muscle contractility and patterning are disrupted to resolve morphological and compositional changes in the ECM during MTJ development. We found that MTJ-specific ECM deposition can be initiated via static loading due to growth; however, it required cyclic loading to develop a mature morphology. Furthermore, the MTJ can mature without the tendon terminating into cartilage. Based on these results, we describe a model wherein MTJ development depends on mechanical loading but not insertion into an enthesis.
Keywords: ECM; MTJ; Muscular dysgenesis (mdg); Musculoskeletal development; Myotendinous junction; Tbx3; Type XXII collagen; Ulnar-mammary syndrome.
Copyright © 2023 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare no competing interests.
Figures


Similar articles
-
Engineering interfacial tissues: The myotendinous junction.APL Bioeng. 2024 Jun 3;8(2):021505. doi: 10.1063/5.0189221. eCollection 2024 Jun. APL Bioeng. 2024. PMID: 38841690 Free PMC article. Review.
-
Preparation of decellularized biphasic hierarchical myotendinous junction extracellular matrix for muscle regeneration.Acta Biomater. 2018 Mar 1;68:15-28. doi: 10.1016/j.actbio.2017.12.035. Epub 2017 Dec 30. Acta Biomater. 2018. PMID: 29294376
-
Larger interface area at the human myotendinous junction in type 1 compared with type 2 muscle fibers.Scand J Med Sci Sports. 2023 Feb;33(2):136-145. doi: 10.1111/sms.14246. Epub 2022 Oct 28. Scand J Med Sci Sports. 2023. PMID: 36226768 Free PMC article.
-
An engineered in vitro model of the human myotendinous junction.Acta Biomater. 2024 May;180:279-294. doi: 10.1016/j.actbio.2024.04.007. Epub 2024 Apr 10. Acta Biomater. 2024. PMID: 38604466 Free PMC article.
-
Role of extracellular matrix in adaptation of tendon and skeletal muscle to mechanical loading.Physiol Rev. 2004 Apr;84(2):649-98. doi: 10.1152/physrev.00031.2003. Physiol Rev. 2004. PMID: 15044685 Review.
Cited by
-
Engineering interfacial tissues: The myotendinous junction.APL Bioeng. 2024 Jun 3;8(2):021505. doi: 10.1063/5.0189221. eCollection 2024 Jun. APL Bioeng. 2024. PMID: 38841690 Free PMC article. Review.
-
3D-environment and muscle contraction regulate the heterogeneity of myonuclei.Skelet Muscle. 2024 Nov 11;14(1):27. doi: 10.1186/s13395-024-00359-x. Skelet Muscle. 2024. PMID: 39529179 Free PMC article.
-
Extracellular matrix deposition precedes muscle-tendon integration during murine forelimb morphogenesis.Commun Biol. 2025 Aug 12;8(1):1202. doi: 10.1038/s42003-025-08653-0. Commun Biol. 2025. PMID: 40796656 Free PMC article.
-
Lack of skeletal muscle contraction disrupts fibrous tissue morphogenesis in the developing murine knee.J Orthop Res. 2023 Oct;41(10):2305-2314. doi: 10.1002/jor.25659. Epub 2023 Jul 26. J Orthop Res. 2023. PMID: 37408453 Free PMC article.
-
Extracellular matrix protein composition dynamically changes during murine forelimb development.iScience. 2024 Jan 9;27(2):108838. doi: 10.1016/j.isci.2024.108838. eCollection 2024 Feb 16. iScience. 2024. PMID: 38303699 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

