This is a preprint.
Analysis of CDPK1 targets identifies a trafficking adaptor complex that regulates microneme exocytosis in Toxoplasma
- PMID: 36712004
- PMCID: PMC9882037
- DOI: 10.1101/2023.01.11.523553
Analysis of CDPK1 targets identifies a trafficking adaptor complex that regulates microneme exocytosis in Toxoplasma
Update in
-
Analysis of CDPK1 targets identifies a trafficking adaptor complex that regulates microneme exocytosis in Toxoplasma.Elife. 2023 Nov 7;12:RP85654. doi: 10.7554/eLife.85654. Elife. 2023. PMID: 37933960 Free PMC article.
Abstract
Apicomplexan parasites use Ca2+-regulated exocytosis to secrete essential virulence factors from specialized organelles called micronemes. Ca2+-dependent protein kinases (CDPKs) are required for microneme exocytosis; however, the molecular events that regulate trafficking and fusion of micronemes with the plasma membrane remain unresolved. Here, we combine sub-minute resolution phosphoproteomics and bio-orthogonal labeling of kinase substrates in Toxoplasma gondii to identify 163 proteins phosphorylated in a CDPK1-dependent manner. In addition to known regulators of secretion, we identify uncharacterized targets with predicted functions across signaling, gene expression, trafficking, metabolism, and ion homeostasis. One of the CDPK1 targets is a putative HOOK activating adaptor. In other eukaryotes, HOOK homologs form the FHF complex with FTS and FHIP to activate dynein-mediated trafficking of endosomes along microtubules. We show the FHF complex is partially conserved in T. gondii, consisting of HOOK, an FTS homolog, and two parasite-specific proteins (TGGT1_306920 and TGGT1_316650). CDPK1 kinase activity and HOOK are required for the rapid apical trafficking of micronemes as parasites initiate motility. Moreover, parasites lacking HOOK or FTS display impaired microneme protein secretion, leading to a block in the invasion of host cells. Taken together, our work provides a comprehensive catalog of CDPK1 targets and reveals how vesicular trafficking has been tuned to support a parasitic lifestyle.
Conflict of interest statement
COMPETING INTERESTS The authors declare no competing interests.
Figures




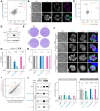
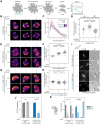

References
-
- Alam MM, Solyakov L, Bottrill AR, Flueck C, Siddiqui FA, Singh S, Mistry S, Viskaduraki M, Lee K, Hopp CS, Chitnis CE, Doerig C, Moon RW, Green JL, Holder AA, Baker DA, Tobin AB. 2015. Phosphoproteomics reveals malaria parasite Protein Kinase G as a signalling hub regulating egress and invasion. Nat Commun 6:7285. - PMC - PubMed
-
- Aquilini E, Cova MM, Mageswaran SK, Dos Santos Pacheco N, Sparvoli D, Penarete-Vargas DM, Najm R, Graindorge A, Suarez C, Maynadier M, Berry-Sterkers L, Urbach S, Fahy PR, Guérin AN, Striepen B, Dubremetz J-F, Chang Y-W, Turkewitz AP, Lebrun M. 2021. An Alveolata secretory machinery adapted to parasite host cell invasion. Nat Microbiol 6:425–434. - PMC - PubMed
-
- Bandini G, Leon DR, Hoppe CM, Zhang Y, Agop-Nersesian C, Shears MJ, Mahal LK, Routier FH, Costello CE, Samuelson J. 2019. O-Fucosylation of thrombospondin-like repeats is required for processing of microneme protein 2 and for efficient host cell invasion by Toxoplasma gondii tachyzoites. J Biol Chem 294:1967–1983. - PMC - PubMed
-
- Barylyuk K, Koreny L, Ke H, Butterworth S, Crook OM, Lassadi I, Gupta V, Tromer E, Mourier T, Stevens TJ, Breckels LM, Pain A, Lilley KS, Waller RF. 2020. A Comprehensive Subcellular Atlas of the Toxoplasma Proteome via hyperLOPIT Provides Spatial Context for Protein Functions. Cell Host Microbe 28:752–766.e9. - PMC - PubMed
-
- Beck JR, Fung C, Straub KW, Coppens I, Vashisht AA, Wohlschlegel JA, Bradley PJ. 2013. A Toxoplasma palmitoyl acyl transferase and the palmitoylated armadillo repeat protein TgARO govern apical rhoptry tethering and reveal a critical role for the rhoptries in host cell invasion but not egress. PLoS Pathog 9:e1003162. - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
