TRIM72 Alleviates Muscle Inflammation in mdx Mice via Promoting Mitophagy-Mediated NLRP3 Inflammasome Inactivation
- PMID: 36713032
- PMCID: PMC9876702
- DOI: 10.1155/2023/8408574
TRIM72 Alleviates Muscle Inflammation in mdx Mice via Promoting Mitophagy-Mediated NLRP3 Inflammasome Inactivation
Abstract
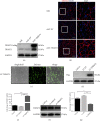
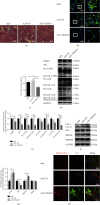
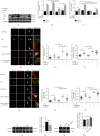
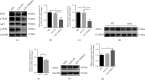
Chronic muscle inflammation exacerbates the pathogenesis of Duchenne muscular dystrophy (DMD), which is characterized by progressive muscle degeneration and weakness. NLRP3 (nucleotide-binding domain and leucine-rich repeat pyrin domain containing 3) inflammasome plays a key role in the inflammatory process, and its abnormal activation leads to a variety of inflammatory or immune diseases. TRIM72 (MG53) is a protective myokine for tissue repair and regeneration. However, little is known about the potential impact of TRIM72 in the crosstalk between mitophagy and inflammatory process of DMD. Here, 10-week-old male mdx mice were injected intramuscularly with adeno-associated virus (AAV-TRIM72) to overexpress TRIM72 protein for 6 weeks. Then, skeletal muscle samples were collected, and relevant parameters were measured by histopathological analysis and molecular biology techniques. C2C12 cell line was transfected with lentivirus (LV-TRIM72) to overexpress or siRNA (si-TRIM72) to suppress the TRIM72 expression for the following experiment. Our data firstly showed that the TRIM72 expression was decreased in skeletal muscles of mdx mice. Then, we observed the increased NLRP3 inflammasome and impaired mitophagy in mdx mice compared with wild type mice. In mdx mice, administration of AAV-TRIM72 alleviated the accumulation of NLRP3 inflammasome and the consequent IL-18 and IL1β maturation by inducing autophagy, while this protective effect was reversed by chloroquine. Mitochondrial reactive oxygen species (mtROS), as a recognized activator for NLRP3 inflammasome, was attenuated by TRIM72 through the induction of mitophagy in C2C12 cells. Additionally, we proposed that the TRIM72 overexpression might promote mitophagy through both the early stage by PI3K-AKT pathway and the late stage by autolysosome fusion. In conclusion, the current study suggests that TRIM72 prevents DMD inflammation via decreasing NLRP3 inflammasomes and enhancing mitophagy. Collectively, our study provides insight into TRIM72 as a promising target for therapeutic intervention for DMD.
Copyright © 2023 Mengli Wu et al.
Conflict of interest statement
All authors declare that there are no competing financial interests exist.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

