A randomized controlled trial to evaluate outcomes with Aggrenox in patients with SARS-CoV-2 infection
- PMID: 36716303
- PMCID: PMC9886260
- DOI: 10.1371/journal.pone.0274243
A randomized controlled trial to evaluate outcomes with Aggrenox in patients with SARS-CoV-2 infection
Abstract
Background: Coronavirus disease 2019 (COVID-19) is an immunoinflammatory and hypercoagulable state that contributes to respiratory distress, multi-organ dysfunction, and mortality. Dipyridamole, by increasing extracellular adenosine, has been postulated to be protective for COVID-19 patients through its immunosuppressive, anti-inflammatory, anti-coagulant, vasodilatory, and anti-viral actions. Likewise, low-dose aspirin has also demonstrated protective effects for COVID-19 patients. This study evaluated the effect of these two drugs formulated together as Aggrenox in hospitalized COVID-19 patients.
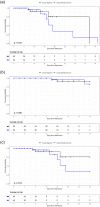
Methods: In an open-label, single site randomized controlled trial (RCT), hospitalized COVID-19 patients were assigned to adjunctive Aggrenox (Dipyridamole ER 200mg/ Aspirin 25mg orally/enterally) with standard of care treatment compared to standard of care treatment alone. Primary endpoint was illness severity according to changes on the eight-point COVID ordinal scale, with levels of 1 to 8 where higher scores represent worse illness. Secondary endpoints included all-cause mortality and respiratory failure. Outcomes were measured through days 14, 28, and/or hospital discharge.
Results: From October 1, 2020 to April 30, 2021, a total of 98 patients, who had a median [IQR] age of 57 [47, 62] years and were 53.1% (n = 52) female, were randomized equally between study groups (n = 49 Aggrenox plus standard of care versus n = 49 standard of care alone). No clinically significant differences were found between those who received adjunctive Aggrenox and the control group in terms of illness severity (COVID ordinal scale) at days 14 and 28. The overall mortality through day 28 was 6.1% (3 patients, n = 49) in the Aggrenox group and 10.2% (5 patients, n = 49) in the control group (OR [95% CI]: 0.40 [0.04, 4.01], p = 0.44). Respiratory failure through day 28 occurred in 4 (8.3%, n = 48) patients in the Aggrenox group and 7 (14.6%, n = 48) patients in the standard of care group (OR [95% CI]: 0.21 [0.02, 2.56], p = 0.22). A larger decrease in the platelet count and blood glucose levels, and larger increase in creatinine and sodium levels within the first 7 days of hospital admission were each independent predictors of 28-day mortality (p < 0.05).
Conclusion: In this study of hospitalized patients with COVID-19, while the outcomes of COVID illness severity, odds of mortality, and chance of respiratory failure were better in the Aggrenox group compared to standard of care alone, the data did not reach statistical significance to support the standard use of adjuvant Aggrenox in such patients.
Copyright: © 2023 Singla et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
References
-
- WHO. Archived: WHO Timeline—COVID-19. 2020. https://www.who.int/news/item/27-04-2020-who-timeline---covid-19 (accessed September 28, 2021.
-
- Marconi VC, Ramanan AV, de Bono S, et al. Efficacy and safety of baricitinib for the treatment of hospitalised adults with COVID-19 (COV-BARRIER): a randomised, double-blind, parallel-group, placebo-controlled phase 3 trial. Lancet Respir Med 2021; 9(12): 1407–18. doi: 10.1016/S2213-2600(21)00331-3 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous