Morphological and sensorimotor phenotypes in a zebrafish CHARGE syndrome model are domain-dependent
- PMID: 36717082
- PMCID: PMC10242184
- DOI: 10.1111/gbb.12839
Morphological and sensorimotor phenotypes in a zebrafish CHARGE syndrome model are domain-dependent
Abstract
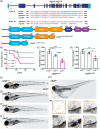
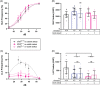
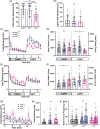
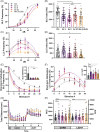
CHARGE syndrome is a heterogeneous disorder characterized by a spectrum of defects affecting multiple tissues and behavioral difficulties such as autism, attention-deficit/hyperactivity disorder, obsessive-compulsive disorder, anxiety, and sensory deficits. Most CHARGE cases arise from de novo, loss-of-function mutations in chromodomain-helicase-DNA-binding-protein-7 (CHD7). CHD7 is required for processes such as neuronal differentiation and neural crest cell migration, but how CHD7 affects neural circuit function to regulate behavior is unclear. To investigate the pathophysiology of behavioral symptoms in CHARGE, we established a mutant chd7 zebrafish line that recapitulates multiple CHARGE phenotypes including ear, cardiac, and craniofacial defects. Using a panel of behavioral assays, we found that chd7 mutants have specific auditory and visual behavior deficits that are independent of defects in sensory structures. Mauthner cell-dependent short-latency acoustic startle responses are normal in chd7 mutants, while Mauthner-independent long-latency responses are reduced. Responses to sudden decreases in light are also reduced in mutants, while responses to sudden increases in light are normal, suggesting that the retinal OFF pathway may be affected. Furthermore, by analyzing multiple chd7 alleles we observed that the penetrance of morphological and behavioral phenotypes is influenced by genetic background but that it also depends on the mutation location, with a chromodomain mutation causing the highest penetrance. This pattern is consistent with analysis of a CHARGE patient dataset in which symptom penetrance was highest in subjects with mutations in the CHD7 chromodomains. These results provide new insight into the heterogeneity of CHARGE and will inform future work to define CHD7-dependent neurobehavioral mechanisms.
Keywords: CHARGE syndrome; CRISPR/Cas9; behavior; chd7; zebrafish.
© 2023 The Authors. Genes, Brain and Behavior published by International Behavioural and Neural Genetics Society and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no competing or financial interests.
Figures

References
-
- Verloes A. Updated diagnostic criteria for CHARGE syndrome: a proposal. Am J Med Genet A. 2005;133A:306‐308. - PubMed
-
- Blake KD, Hartshorne TS, Lawand C, Dailor AN, Thelin JW. Cranial nerve manifestations in CHARGE syndrome. Am J Med Genet A. 2008;146A:585‐592. - PubMed
-
- Blake KD, Salem‐Hartshorne N, Daoud MA, Gradstein J. Adolescent and adult issues in CHARGE syndrome. Clin Pediatr. 2005;44:151‐159. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

