Outcome of COVID-19 in hospitalised immunocompromised patients: An analysis of the WHO ISARIC CCP-UK prospective cohort study
- PMID: 36719907
- PMCID: PMC9928075
- DOI: 10.1371/journal.pmed.1004086
Outcome of COVID-19 in hospitalised immunocompromised patients: An analysis of the WHO ISARIC CCP-UK prospective cohort study
Abstract
Background: Immunocompromised patients may be at higher risk of mortality if hospitalised with Coronavirus Disease 2019 (COVID-19) compared with immunocompetent patients. However, previous studies have been contradictory. We aimed to determine whether immunocompromised patients were at greater risk of in-hospital death and how this risk changed over the pandemic.
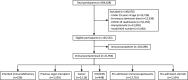
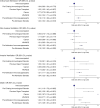
Methods and findings: We included patients > = 19 years with symptomatic community-acquired COVID-19 recruited to the ISARIC WHO Clinical Characterisation Protocol UK prospective cohort study. We defined immunocompromise as immunosuppressant medication preadmission, cancer treatment, organ transplant, HIV, or congenital immunodeficiency. We used logistic regression to compare the risk of death in both groups, adjusting for age, sex, deprivation, ethnicity, vaccination, and comorbidities. We used Bayesian logistic regression to explore mortality over time. Between 17 January 2020 and 28 February 2022, we recruited 156,552 eligible patients, of whom 21,954 (14%) were immunocompromised. In total, 29% (n = 6,499) of immunocompromised and 21% (n = 28,608) of immunocompetent patients died in hospital. The odds of in-hospital mortality were elevated for immunocompromised patients (adjusted OR 1.44, 95% CI [1.39, 1.50], p < 0.001). Not all immunocompromising conditions had the same risk, for example, patients on active cancer treatment were less likely to have their care escalated to intensive care (adjusted OR 0.77, 95% CI [0.7, 0.85], p < 0.001) or ventilation (adjusted OR 0.65, 95% CI [0.56, 0.76], p < 0.001). However, cancer patients were more likely to die (adjusted OR 2.0, 95% CI [1.87, 2.15], p < 0.001). Analyses were adjusted for age, sex, socioeconomic deprivation, comorbidities, and vaccination status. As the pandemic progressed, in-hospital mortality reduced more slowly for immunocompromised patients than for immunocompetent patients. This was particularly evident with increasing age: the probability of the reduction in hospital mortality being less for immunocompromised patients aged 50 to 69 years was 88% for men and 83% for women, and for those >80 years was 99% for men and 98% for women. The study is limited by a lack of detailed drug data prior to admission, including steroid doses, meaning that we may have incorrectly categorised some immunocompromised patients as immunocompetent.
Conclusions: Immunocompromised patients remain at elevated risk of death from COVID-19. Targeted measures such as additional vaccine doses, monoclonal antibodies, and nonpharmaceutical preventive interventions should be continually encouraged for this patient group.
Trial registration: ISRCTN 66726260.
Copyright: © 2023 Turtle et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: LT declares a lecture fee paid to his institution from Eisai ltd. CP reports research grants from Pfizer, Daiichi Sankyo and Seagen, consulting fees from Pfizer, Roche, Daiichi Sankyo, Novartis, Exact sciences, Gilead, SeaGen and Eli Lilly, and payment for lectures from Pfizer, Novartis and Eisai ltd. PO declares fees from Affnivax, Oxford Immunotech, Nestle, Pfizer and Janessen for consulting or chairing a symposium, paid to Imperial College. MGS is chair of the infectious disease advisory board for Integrum Scientific, director of MedEx solutions ltd, participated in the DSMB for Pfizer COVID-19 vaccine trials, and was donated an investigational medicinal product by Chiesi Farmaceutici S.p.A. MGS and PJMO sat on HMG UK New Emerging Respiratory Virus Threats Advisory Group (NERVTAG). MGS sat on HMG UK Scientific Advisory Group for Emergencies (SAGE), COVID-19 Response from March 2020 to March 2022.
Figures

References
-
- Thwaites RS, Sanchez Sevilla Uruchurtu A, Siggins MK, Liew F, Russell CD, Moore SC, et al. Inflammatory profiles across the spectrum of disease reveal a distinct role for GM-CSF in severe COVID-19. Sci Immunol. 2021;6(57). Epub 2021/03/12. doi: 10.1126/sciimmunol.abg9873 [pii] ; PubMed Central PMCID: PMC8128298. - DOI - PMC - PubMed
-
- Hoepel W, Chen HJ, Geyer CE, Allahverdiyeva S, Manz XD, de Taeye SW, et al. High titers and low fucosylation of early human anti-SARS-CoV-2 IgG promote inflammation by alveolar macrophages. Sci Transl Med. 2021;13(596). Epub 2021/05/13. scitranslmed.abf8654 [pii] doi: 10.1126/scitranslmed.abf8654 ; PubMed Central PMCID: PMC8158960. - DOI - PMC - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

