Temporal evolution of new T1-weighted hypo-intense lesions and central brain atrophy in patients with a first clinical demyelinating event treated with subcutaneous interferon β-1a
- PMID: 36723685
- PMCID: PMC10025187
- DOI: 10.1007/s00415-022-11554-5
Temporal evolution of new T1-weighted hypo-intense lesions and central brain atrophy in patients with a first clinical demyelinating event treated with subcutaneous interferon β-1a
Abstract
Objective: Evaluate the effect of subcutaneous interferon β-1a (sc IFN β-1a) versus placebo on the evolution of T1-weighted MRI lesions and central brain atrophy in in patients with a first clinical demyelinating event (FCDE).
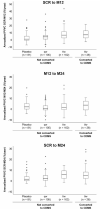
Methods: Post hoc analysis of baseline-to-24 month MRI data from patients with an FCDE who received sc IFN β-1a 44 μg once- (qw) or three-times-weekly (tiw), or placebo, in REFLEX. Patients were grouped according to treatment regimen or conversion to clinically definite MS (CDMS) status. The intensity of new lesions on unenhanced T1-weighted images was classified as T1 iso- or hypo-intense (black holes) and percentage ventricular volume change (PVVC) was assessed throughout the study.
Results: In patients not converting to CDMS, sc IFN β-1a tiw or qw, versus placebo, reduced the overall number of new lesions (P < 0.001 and P = 0.005) and new T1 iso-intense lesions (P < 0.001 and P = 0.002) after 24 months; only sc IFN β-1a tiw was associated with fewer T1 hypo-intense lesions versus placebo (P < 0.001). PVVC findings in patients treated with sc IFN β-1a suggested pseudo-atrophy that was ~ fivefold greater versus placebo in the first year of treatment (placebo 1.11%; qw 4.28%; tiw 6.76%; P < 001); similar findings were apparent for non-converting patients.
Conclusions: In patients with an FCDE, treatment with sc IFN β-1a tiw for 24 months reduced the number of new lesions evolving into black holes.
Keywords: Black-hole lesions; Brain atrophy; First clinical demyelinating event; Interferon β-1a; Lesion evolution; White matter tracts.
© 2023. The Author(s).
Conflict of interest statement
HV has received research support from Merck, Novartis, Pfizer, and Teva, consulting fees from Merck, and speaker honoraria from Novartis; all funds were paid to his institution. GJN has received personal compensation for activities with Bayer and research support from Merck. AS is an employee of Merck Healthcare KGaA, Darmstadt, Germany. DJ is an employee of Merck Serono Ltd, Feltham, UK (an affiliate of Merck KGaA). MPS has received consulting fees from Biogen, Genzyme, GeNeuro, MedDay, Merck, Novartis, Roche, and Teva. BMJU has received consultancy fees from Biogen, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva. AV has received research support from Merck. GC has received consulting fees from Bayer, Biogen, Merck, Novartis, Receptos, Roche/Genentech, Sanofi-Aventis, and Teva Pharmaceutical Industries Ltd; lecture fees from Bayer, Biogen, Merck, Novartis, Sanofi-Aventis, Serono Symposia International Foundation, and Teva Pharmaceutical Industries Ltd; and trial grant support from Bayer, Biogen, Merck, Novartis, Receptos, Roche/Genentech, Sanofi-Aventis, and Teva Pharmaceutical Industries Ltd. LK’s institution (University Hospital Basel, University of Basel) has received in the last 3 years and used exclusively for research support: steering committee, advisory board, and consultancy fees (Actelion [Janssen/JandJ], Addex, Bayer, Biogen, Biotica, Genzyme, Lilly, Merck, Mitsubishi, Novartis, Ono Pharma, Pfizer, Receptos, Sanofi, Santhera, Siemens, Teva, UCB, and Xenoport); speaker fees (Bayer, Biogen, Merck, Novartis, Sanofi, and Teva); support of educational activities (Bayer, Biogen, CSL Behring, Genzyme, Merck, Novartis, Sanofi, and Teva); license fees for Neurostatus products; and grants (Bayer, Biogen, European Union, Merck, Novartis, Roche Research Foundation, Swiss MS Society, and Swiss National Research Foundation). NDeS is a consultant for Biogen, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva; has grants or grants pending from FISM and Novartis, is on the speakers’ bureaus of Biogen, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva; and has received travel funds from Merck, Novartis, Roche, Sanofi-Genzyme, and Teva. FB is supported by the NIHR Biomedical Research Centre at UCLH and is a consultant to Biogen, Combinostics, IXICO, Merck, and Roche. MB, MLdeV, and BCAT have nothing to disclose.
Figures


References
-
- Kinkel RP, Kollman C, O'Connor P, et al. IM interferon beta-1a delays definite multiple sclerosis 5 years after a first demyelinating event. Neurology. 2006;66(5):678–684. doi: 10.1212/01.wnl.0000200778.65597.ae. - DOI - PubMed
-
- Kappos L, Freedman MS, Polman CH, et al. Effect of early versus delayed interferon beta-1b treatment on disability after a first clinical event suggestive of multiple sclerosis: a 3-year follow-up analysis of the BENEFIT study. Lancet. 2007;370(9585):389–397. doi: 10.1016/S0140-6736(07)61194-5. - DOI - PubMed
-
- Comi G, De Stefano N, Freedman MS, et al. Comparison of two dosing frequencies of subcutaneous interferon beta-1a in patients with a first clinical demyelinating event suggestive of multiple sclerosis (REFLEX): a phase 3 randomised controlled trial. Lancet Neurol. 2012;11(1):33–41. doi: 10.1016/S1474-4422(11)70262-9. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

