Application of the Latest Advances in Evidence-Based Medicine in Primary Biliary Cholangitis
- PMID: 36729104
- PMCID: PMC9889200
- DOI: 10.14309/ajg.0000000000002070
Application of the Latest Advances in Evidence-Based Medicine in Primary Biliary Cholangitis
Abstract
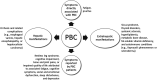
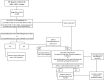
Primary biliary cholangitis (PBC) is a chronic, cholestatic, autoimmune liver disease that can progress to end-stage liver disease and its complications. A previous expert review panel collaborated on a consensus document for gastroenterologists and other healthcare professionals regarding the care of patients with PBC. Subsequently, there have been several recent important developments in the diagnosis, treatment, and monitoring of patients with PBC. These include updates to prognostic models on risk stratification, new noninvasive tools for staging of disease, updates to the appropriate use of and long-term treatment results with obeticholic acid as a second-line treatment, the emerging therapeutic role of fibrates, and the advancement of investigational agents for managing PBC. In this updated expert consensus document, we provide updates on staging, the use of noninvasive prognostic tools, and a treatment algorithm to provide evidence-based and practical tools for clinicians who manage PBC, with the ultimate goal to improve the long-term outcomes for patients with this chronic liver disease.
Copyright © 2022 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of The American College of Gastroenterology.
Conflict of interest statement
Figures

References
-
- Murillo Perez CF, Goet JC, Lammers WJ, et al. Milder disease stage in patients with primary biliary cholangitis over a 44-year period: A changing natural history. Hepatology 2018;67:1920–30. - PubMed
-
- Younossi ZM, Bernstein D, Shiffman ML, et al. Diagnosis and management of primary biliary cholangitis. Am J Gastroenterol 2019;114:48–63. - PubMed
-
- Lindor KD, Bowlus CL, Boyer J, et al. Primary biliary cholangitis: 2021 practice guidance update from the American association for the study of liver diseases. Hepatology 2021;75:1012–3. - PubMed
-
- EASL Clinical Practice Guidelines. The diagnosis and management of patients with primary biliary cholangitis. J Hepatol 2017;67:145–72. - PubMed
-
- Lindor KD, Bowlus CL, Boyer J, et al. Primary biliary cholangitis: 2018 practice guidance from the American association for the study of liver diseases. Hepatology 2019;69:394–419. - PubMed

