Biomarkers: Promising and valuable tools towards diagnosis, prognosis and treatment of Covid-19 and other diseases
- PMID: 36744065
- PMCID: PMC9884646
- DOI: 10.1016/j.heliyon.2023.e13323
Biomarkers: Promising and valuable tools towards diagnosis, prognosis and treatment of Covid-19 and other diseases
Abstract
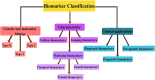
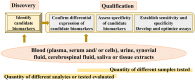
The use of biomarkers as early warning systems in the evaluation of disease risk has increased markedly in the last decade. Biomarkers are indicators of typical biological processes, pathogenic processes, or pharmacological reactions to therapy. The application and identification of biomarkers in the medical and clinical fields have an enormous impact on society. In this review, we discuss the history, various definitions, classifications, characteristics, and discovery of biomarkers. Furthermore, the potential application of biomarkers in the diagnosis, prognosis, and treatment of various diseases over the last decade are reviewed. The present review aims to inspire readers to explore new avenues in biomarker research and development.
Keywords: Biomarker; Cancer; Covid-19; Diagnosis; Diseases; Imaging; Treatment.
© 2023 The Authors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Beasley V.R., Levengood J.M. Elsevier Inc.; 2012. Principles of Ecotoxicology, Veterinary Toxicology; pp. 831–855.
-
- Godfrey A., Vandendriessche B., Bakker J.P., Fitzer‐Attas C., Gujar N., Hobbs M., Liu Q., Northcott C.A., Parks V., Wood W.A. Fit‐for‐purpose biometric monitoring technologies: leveraging the laboratory biomarker experience. Clin. Transl. Sci. 2021;14:62–74. doi: 10.1111/cts.12865. - DOI - PMC - PubMed
-
- Group B.D.W., Atkinson A.J., Jr., Colburn W.A., DeGruttola V.G., DeMets D.L., Downing G.J., Hoth D.F., Oates J.A., Peck C.C., Schooley R.T. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 2001;69:89–95. doi: 10.1067/mcp.2001.113989. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

