Infectivity and Shedding of Mouse Kidney Parvovirus After Oronasal Inoculation of C57BL/6, CD1, and NSG Mice
- PMID: 36744512
- PMCID: PMC9827609
- DOI: 10.30802/AALAS-CM-22-000066
Infectivity and Shedding of Mouse Kidney Parvovirus After Oronasal Inoculation of C57BL/6, CD1, and NSG Mice
Abstract
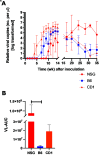
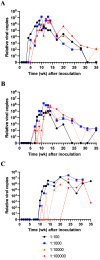
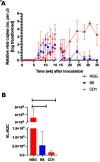
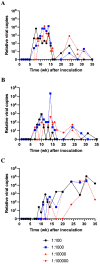
Mouse kidney parvovirus (MKPV), the etiology of murine inclusion body nephropathy, has been identified globally in mice used for research, with an estimated prevalence of 10% in academic colonies. In immunodeficient strains, MKPV causes significant morbidity and mortality, and severe renal pathology. In contrast, in immunocompetent mice, the infection is subclinical and causes minimal pathology. We investigated viral infectivity and shedding in inbred C57BL/6NCrl (B6), outbred Crl:CD1(ICR) (CD1), and highly immunocompromised NOD. Cg - Prkdc scid Il2rg tm1Wjl/SzJ (NSG) mice. Four doses, ranging from 1.16 × 10 3 to 1.16 × 10 6 viral copies per microliter, of an MKPV inoculum were administered oronasally to 3 mice per dose per mouse type. All 3 types (B6, CD1, and NSG) had persistent infection with prolonged shedding in urine and feces. Viral copy number in the urine generally increased over time, while shedding in the feces was more variable. Among the 3 populations, CD1 mice developed viral shedding in urine earliest (4 wk after inoculation) and at higher levels (greater than 1 × 10 7 viral copies per microliter). B6 mice become viruric later (7 wk after inoculation), with lesser virus shed (1 × 10 6 viral copies per microliter or less). In CD1 and B6 mice, peak urine shedding occurred at 11 to 14 wk after inoculation, after which levels gradually declined until 35 wk after inoculation (study endpoint). In contrast, NSG mice did not become viruric until 10 wk after inoculation and continued to shed large amounts of virus (greater than 1 × 107 viral copies per microliter) in urine until the study endpoint. Two commercial immunofluorescent serologic assays failed to detect serum antibodies to MKPV nonstructural protein 1 as late as 58 wk after inoculation, whereas immunohistochemistry of infected renal tissue successfully detected anti-MKPV serum antibodies. These results increase our knowledge of the biology of MKPV and have practical application for development of effective screening programs for this pathogen.
Figures

Similar articles
-
Prevalence and Pathologic Characterization of Mouse Kidney Parvovirus in Sentinel CD1 Mice.Comp Med. 2024 Oct 31;74(5):344-351. doi: 10.30802/AALAS-CM-24-000015. Print 2024 Oct 1. Comp Med. 2024. PMID: 39503174 Free PMC article.
-
Effects of Mouse Kidney Parvovirus on Pharmacokinetics of Chemotherapeutics and the Adenine Model of Chronic Kidney Disease.Comp Med. 2023 Apr 1;73(2):153-172. doi: 10.30802/AALAS-CM-22-000084. Epub 2023 Mar 27. Comp Med. 2023. PMID: 36973002 Free PMC article.
-
Identification of a new strain of mouse kidney parvovirus associated with inclusion body nephropathy in immunocompromised laboratory mice.Emerg Microbes Infect. 2020 Dec;9(1):1814-1823. doi: 10.1080/22221751.2020.1798288. Emerg Microbes Infect. 2020. PMID: 32686622 Free PMC article.
-
Assessing Elimination of Mouse Kidney Parvovirus from Cages by Mechanical Washing.J Am Assoc Lab Anim Sci. 2022 Jan 1;61(1):61-66. doi: 10.30802/AALAS-JAALAS-21-000096. Epub 2021 Dec 17. J Am Assoc Lab Anim Sci. 2022. PMID: 34920766 Free PMC article.
-
Naturally Acquired Mouse Kidney Parvovirus Infection Produces a Persistent Interstitial Nephritis in Immunocompetent Laboratory Mice.Vet Pathol. 2020 Nov;57(6):915-925. doi: 10.1177/0300985820953500. Epub 2020 Oct 5. Vet Pathol. 2020. PMID: 33016243
Cited by
-
Do we still need a canary in the coal mine for laboratory animal facilities? A systematic review of environmental health monitoring versus soiled bedding sentinels.PLoS One. 2024 Dec 5;19(12):e0311840. doi: 10.1371/journal.pone.0311840. eCollection 2024. PLoS One. 2024. PMID: 39636910 Free PMC article.
-
Prevalence and Pathologic Characterization of Mouse Kidney Parvovirus in Sentinel CD1 Mice.Comp Med. 2024 Oct 31;74(5):344-351. doi: 10.30802/AALAS-CM-24-000015. Print 2024 Oct 1. Comp Med. 2024. PMID: 39503174 Free PMC article.
References
-
- Baze WB, Steinbach TJ, Fleetwood ML, Blanchard TW, Barnhart KF, McArthur MJ. 2006. Karyomegaly and intranuclear inclusions in the renal tubules of sentinel ICR mice (Mus musculus). Comp Med 56:435–438. - PubMed
-
- Besselsen DG, Wagner AM, Loganbill JK. 2000. Effect of mouse strain and age on detection of mouse parvovirus 1 by use of serologic testing and polymerase chain reaction analysis. Comp Med 50:498–502. - PubMed
-
- Brownstein DG, Smith AL, Jacoby RO, Johnson EA, Hansen G, Tattersall P. 1991. Pathogenesis of infection with a virulent allotropic variant of minute virus of mice and regulation by host genotype. Lab Invest 65:357–364. - PubMed
-
- Edmondson EF, Hsieh WT, Kramer JA, Breed MW, Roelke-Parker ME, Stephens-Devalle J, Pate NM, Bassel LL, Hollingshead MG, Karim BO, Butcher DO, Warner AC, Nagashima K, Gulani J. 2020. Naturally acquired mouse kidney parvovirus infection produces a persistent interstitial nephritis in immunocompetent laboratory mice. Vet Pathol 57:915–925. 10.1177/0300985820953500. - DOI - PubMed
-
- Ge Z, Carrasco SE, Feng Y, Bakthavatchalu V, Annamalai D, Kramer R, Muthupalani S, Fox JG. 2020. Identification of a new strain of mouse kidney parvovirus associated with inclusion body nephropathy in immunocompromised laboratory mice. Emerg Microbes Infect 9:1814–1823. 10.1080/22221751.2020.1798288. - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

