Infection burdens and virulence under heat stress: ecological and evolutionary considerations
- PMID: 36744570
- PMCID: PMC9900716
- DOI: 10.1098/rstb.2022.0018
Infection burdens and virulence under heat stress: ecological and evolutionary considerations
Abstract
As a result of global change, hosts and parasites (including pathogens) are experiencing shifts in their thermal environment. Despite the importance of heat stress tolerance for host population persistence, infection by parasites can impair a host's ability to cope with heat. Host-parasite eco-evolutionary dynamics will be affected if infection reduces host performance during heating. Theory predicts that within-host parasite burden (replication rate or number of infecting parasites per host), a key component of parasite fitness, should correlate positively with virulence-the harm caused to hosts during infection. Surprisingly, however, the relationship between within-host parasite burden and virulence during heating is often weak. Here, we describe the current evidence for the link between within-host parasite burden and host heat stress tolerance. We consider the biology of host-parasite systems that may explain the weak or absent link between these two important host and parasite traits during hot conditions. The processes that mediate the relationship between parasite burden and host fitness will be fundamental in ecological and evolutionary responses of host and parasites in a warming world. This article is part of the theme issue 'Infectious disease ecology and evolution in a changing world'.
Keywords: disease burden; pathogen evolution; thermal tolerance; trade-off; transmission; virulence.
Figures
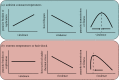
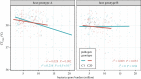
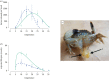
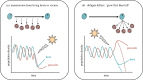
Similar articles
-
Multispecies interactions and the community context of the evolution of virulence.Proc Biol Sci. 2024 Sep;291(2031):20240991. doi: 10.1098/rspb.2024.0991. Epub 2024 Sep 25. Proc Biol Sci. 2024. PMID: 39317313 Free PMC article.
-
Bidirectional interactions between host social behaviour and parasites arise through ecological and evolutionary processes.Parasitology. 2021 Mar;148(3):274-288. doi: 10.1017/S0031182020002048. Epub 2020 Oct 23. Parasitology. 2021. PMID: 33092680 Free PMC article.
-
Growth rate, transmission mode and virulence in human pathogens.Philos Trans R Soc Lond B Biol Sci. 2017 May 5;372(1719):20160094. doi: 10.1098/rstb.2016.0094. Philos Trans R Soc Lond B Biol Sci. 2017. PMID: 28289261 Free PMC article.
-
How Individual Variation in Host Tolerance Affects Competence to Transmit Parasites.Physiol Biochem Zool. 2019 Jan/Feb;92(1):49-57. doi: 10.1086/701169. Physiol Biochem Zool. 2019. PMID: 30481116 Review.
-
How do fluctuating ecological dynamics impact the evolution of hosts and parasites?Philos Trans R Soc Lond B Biol Sci. 2023 Mar 27;378(1873):20220006. doi: 10.1098/rstb.2022.0006. Epub 2023 Feb 6. Philos Trans R Soc Lond B Biol Sci. 2023. PMID: 36744565 Free PMC article. Review.
Cited by
-
Would global warming bring an increase of invertebrate-associated cutaneous invasive fungal infections?mBio. 2025 Mar 12;16(3):e0344724. doi: 10.1128/mbio.03447-24. Epub 2025 Feb 5. mBio. 2025. PMID: 39907444 Free PMC article. Review.
-
Adaptive evolution of stress response genes in parasites aligns with host niche diversity.BMC Biol. 2025 Jan 13;23(1):10. doi: 10.1186/s12915-024-02091-w. BMC Biol. 2025. PMID: 39800686 Free PMC article.
-
Infectious disease ecology and evolution in a changing world.Philos Trans R Soc Lond B Biol Sci. 2023 Mar 27;378(1873):20220002. doi: 10.1098/rstb.2022.0002. Epub 2023 Feb 6. Philos Trans R Soc Lond B Biol Sci. 2023. PMID: 36744560 Free PMC article. No abstract available.
-
Warming alters life-history traits and competition in a phage community.Appl Environ Microbiol. 2024 May 21;90(5):e0028624. doi: 10.1128/aem.00286-24. Epub 2024 Apr 16. Appl Environ Microbiol. 2024. PMID: 38624196 Free PMC article.
-
Editorial: Parasitism: the good, the bad and the ugly.Front Vet Sci. 2023 Oct 17;10:1304206. doi: 10.3389/fvets.2023.1304206. eCollection 2023. Front Vet Sci. 2023. PMID: 37915945 Free PMC article. No abstract available.
References
-
- Kingsolver JG, Diamond SE, Buckley LB. 2013. Heat stress and the fitness consequences of climate change for terrestrial ectotherms. Funct. Ecol 27, 1415-1423. (10.1111/1365-2435.12145) - DOI
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Research Materials

