Application of microfluidic chips in the simulation of the urinary system microenvironment
- PMID: 36747584
- PMCID: PMC9898763
- DOI: 10.1016/j.mtbio.2023.100553
Application of microfluidic chips in the simulation of the urinary system microenvironment
Abstract
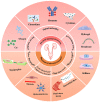
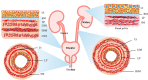
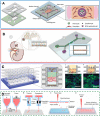
The urinary system, comprising the kidneys, ureters, bladder, and urethra, has a unique mechanical and fluid microenvironment, which is essential to the urinary system growth and development. Microfluidic models, based on micromachining and tissue engineering technology, can integrate pathophysiological characteristics, maintain cell-cell and cell-extracellular matrix interactions, and accurately simulate the vital characteristics of human tissue microenvironments. Additionally, these models facilitate improved visualization and integration and meet the requirements of the laminar flow environment of the urinary system. However, several challenges continue to impede the development of a tissue microenvironment with controllable conditions closely resemble physiological conditions. In this review, we describe the biochemical and physical microenvironment of the urinary system and explore the feasibility of microfluidic technology in simulating the urinary microenvironment and pathophysiological characteristics in vitro. Moreover, we summarize the current research progress on adapting microfluidic chips for constructing the urinary microenvironment. Finally, we discuss the current challenges and suggest directions for future development and application of microfluidic technology in constructing the urinary microenvironment in vitro.
Keywords: Microfluidic chip; Tissue engineering; Urinary microenvironment; Urinary system.
© 2023 The Authors. Published by Elsevier Ltd.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





Similar articles
-
[Chondrocyte microenvironment and application of microfluidic chips in constructing chondrocyte microenvironment].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2014 Jan;28(1):105-8. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2014. PMID: 24693790 Review. Chinese.
-
Advances of 3D Cell Co-Culture Technology Based on Microfluidic Chips.Biosensors (Basel). 2024 Jul 10;14(7):336. doi: 10.3390/bios14070336. Biosensors (Basel). 2024. PMID: 39056612 Free PMC article. Review.
-
Microfluidic Organs-on-a-Chip for Modeling Human Infectious Diseases.Acc Chem Res. 2021 Sep 21;54(18):3550-3562. doi: 10.1021/acs.accounts.1c00411. Epub 2021 Aug 29. Acc Chem Res. 2021. PMID: 34459199
-
Applications of microfluidic chip technology in microvascular thrombosis research.Mikrochim Acta. 2025 May 24;192(6):371. doi: 10.1007/s00604-025-07239-1. Mikrochim Acta. 2025. PMID: 40413353 Review.
-
[Applications of microfluidic paper-based chips in environmental analysis and detection].Se Pu. 2021 Aug;39(8):802-815. doi: 10.3724/SP.J.1123.2020.09004. Se Pu. 2021. PMID: 34212581 Free PMC article. Chinese.
Cited by
-
Harnessing the power of artificial intelligence for human living organoid research.Bioact Mater. 2024 Aug 30;42:140-164. doi: 10.1016/j.bioactmat.2024.08.027. eCollection 2024 Dec. Bioact Mater. 2024. PMID: 39280585 Free PMC article. Review.
-
Porous Structural Microfluidic Device for Biomedical Diagnosis: A Review.Micromachines (Basel). 2023 Feb 26;14(3):547. doi: 10.3390/mi14030547. Micromachines (Basel). 2023. PMID: 36984956 Free PMC article. Review.
References
-
- Jones-Freeman B., Chonwerawong M., Marcelino V.R., Deshpande A.V., Forster S.C., Mr S. The microbiome and host mucosal interactions in urinary tract diseases. Mucosal Immunol. 2021;14(4):779–792. - PubMed
-
- Makkar H., Zhou Y., Tan K.S., Lim Ct S.G. Modelling crevicular fluid flow and host-Oral microbiome interactions in a gingival crevice-on-Chip. Adv Healthc Mater. 2022;17 - PubMed
Publication types
LinkOut - more resources
Full Text Sources

