The role of amphiregulin in ovarian function and disease
- PMID: 36749397
- PMCID: PMC11071807
- DOI: 10.1007/s00018-023-04709-8
The role of amphiregulin in ovarian function and disease
Abstract
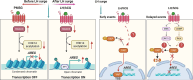
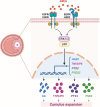
Amphiregulin (AREG) is an epidermal growth factor (EGF)-like growth factor that binds exclusively to the EGF receptor (EGFR). Treatment with luteinizing hormone (LH) and/or human chorionic gonadotropin dramatically induces the expression of AREG in the granulosa cells of the preovulatory follicle. In addition, AREG is the most abundant EGFR ligand in human follicular fluid. Therefore, AREG is considered a predominant propagator that mediates LH surge-regulated ovarian functions in an autocrine and/or paracrine manner. In addition to the well-characterized stimulatory effect of LH on AREG expression, recent studies discovered that several local factors and epigenetic modifications participate in the regulation of ovarian AREG expression. Moreover, aberrant expression of AREG has recently been reported to contribute to the pathogenesis of several ovarian diseases, such as ovarian hyperstimulation syndrome, polycystic ovary syndrome, and epithelial ovarian cancer. Furthermore, increasing evidence has elucidated new applications of AREG in assisted reproductive technology. Collectively, these studies highlight the importance of AREG in female reproductive health and disease. Understanding the normal and pathological roles of AREG and elucidating the molecular and cellular mechanisms of AREG regulation of ovarian functions will inform innovative approaches for fertility regulation and the prevention and treatment of ovarian diseases. Therefore, this review summarizes the functional roles of AREG in ovarian function and disease.
Keywords: Amphiregulin; Assisted reproductive technology; Granulosa cells; Ovary.
© 2023. The Author(s), under exclusive licence to Springer Nature Switzerland AG.
Conflict of interest statement
The authors have not disclosed any competing interests.
Figures




References
-
- Shoyab M, McDonald VL, Bradley JG, Todaro GJ. Amphiregulin: a bifunctional growth-modulating glycoprotein produced by the phorbol 12-myristate 13-acetate-treated human breast adenocarcinoma cell line MCF-7. Proc Natl Acad Sci USA. 1988;85:6528–6532. doi: 10.1073/pnas.85.17.6528. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

