Limbic progesterone receptors regulate spatial memory
- PMID: 36750584
- PMCID: PMC9905062
- DOI: 10.1038/s41598-023-29100-2
Limbic progesterone receptors regulate spatial memory
Abstract
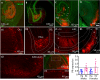
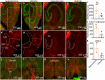
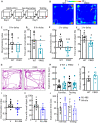
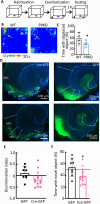
Progesterone and its receptors (PRs) participate in mating and reproduction, but their role in spatial declarative memory is not understood. Male mice expressed PRs, predominately in excitatory neurons, in brain regions that support spatial memory, such as the hippocampus and entorhinal cortex (EC). Furthermore, segesterone, a specific PR agonist, activates neurons in both the EC and hippocampus. We assessed the contribution of PRs in promoting spatial and non-spatial cognitive learning in male mice by examining the performance of mice lacking this receptor (PRKO), in novel object recognition, object placement, Y-maze alternation, and Morris-Water Maze (MWM) tasks. In the recognition test, the PRKO mice preferred the familiar object over the novel object. A similar preference for the familiar object was also seen following the EC-specific deletion of PRs. PRKO mice were also unable to recognize the change in object position. We confirmed deficits in spatial memory of PRKO mice by testing them on the Y-maze forced alternation and MWM tasks; PR deletion affected animal's performance in both these tasks. In contrast to spatial tasks, PR removal did not alter the response to fear conditioning. These studies provide novel insights into the role of PRs in facilitating spatial, declarative memory in males, which may help with finding reproductive partners.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Ogawa S, Olazabal U, Parhar I, Pfaff D. Effects of intrahypothalamic administration of antisense DNA for progesterone receptor mRNA on reproductive behavior and progesterone receptor immunoreactivity in female rat. J. Neurosci. 1994;14:1766–1774. doi: 10.1523/jneurosci.14-03-01766.1994. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

