Mutated HRAS Activates YAP1-AXL Signaling to Drive Metastasis of Head and Neck Cancer
- PMID: 36753744
- PMCID: PMC10073343
- DOI: 10.1158/0008-5472.CAN-22-2586
Mutated HRAS Activates YAP1-AXL Signaling to Drive Metastasis of Head and Neck Cancer
Abstract
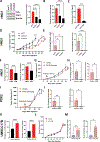
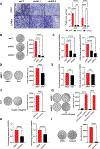
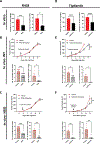
The survival rate for patients with head and neck cancer (HNC) diagnosed with cervical lymph node (cLN) or distant metastasis is low. Genomic alterations in the HRAS oncogene are associated with advanced tumor stage and metastasis in HNC. Elucidation of the molecular mechanisms by which mutated HRAS (HRASmut) facilitates HNC metastasis could lead to improved treatment options for patients. Here, we examined metastasis driven by mutant HRAS in vitro and in vivo using HRASmut human HNC cell lines, patient-derived xenografts, and a novel HRASmut syngeneic model. Genetic and pharmacological manipulations indicated that HRASmut was sufficient to drive invasion in vitro and metastasis in vivo. Targeted proteomic analysis showed that HRASmut promoted AXL expression via suppressing the Hippo pathway and stabilizing YAP1 activity. Pharmacological blockade of HRAS signaling with the farnesyltransferase inhibitor tipifarnib activated the Hippo pathway and reduced the nuclear export of YAP1, thus suppressing YAP1-mediated AXL expression and metastasis. AXL was required for HRASmut cells to migrate and invade in vitro and to form regional cLN and lung metastases in vivo. In addition, AXL-depleted HRASmut tumors displayed reduced lymphatic and vascular angiogenesis in the primary tumor. Tipifarnib treatment also regulated AXL expression and attenuated VEGFA and VEGFC expression, thus regulating tumor-induced vascular formation and metastasis. Our results indicate that YAP1 and AXL are crucial factors for HRASmut-induced metastasis and that tipifarnib treatment can limit the metastasis of HNC tumors with HRAS mutations by enhancing YAP1 cytoplasmic sequestration and downregulating AXL expression.
Significance: Mutant HRAS drives metastasis of head and neck cancer by switching off the Hippo pathway to activate the YAP1-AXL axis and to stimulate lymphovascular angiogenesis.
©2023 American Association for Cancer Research.
Conflict of interest statement
Conflict of Interest
ME reported a grant from Kura Oncology.GG is an employee of BerGen Bio. JRG is the co-inventor of a decoy oligonucleotide targeting STAT3 that has been licensed by BluedotBio. JSG has received financial support from Kura Oncology, and he is consultant for io9, Domain Therapeutics, Pangea Therapeutics, and founder of Kadima pharmaceuticals, outside the submitted work. All others authors declare no conflicts of interest.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

