A subset of antibodies targeting citrullinated proteins confers protection from rheumatoid arthritis
- PMID: 36754962
- PMCID: PMC9908943
- DOI: 10.1038/s41467-023-36257-x
A subset of antibodies targeting citrullinated proteins confers protection from rheumatoid arthritis
Abstract
Although elevated levels of anti-citrullinated protein antibodies (ACPAs) are a hallmark of rheumatoid arthritis (RA), the in vivo functions of these antibodies remain unclear. Here, we have expressed monoclonal ACPAs derived from patients with RA, and analyzed their functions in mice, as well as their specificities. None of the ACPAs showed arthritogenicity nor induced pain-associated behavior in mice. However, one of the antibodies, clone E4, protected mice from antibody-induced arthritis. E4 showed a binding pattern restricted to skin, macrophages and dendritic cells in lymphoid tissue, and cartilage derived from mouse and human arthritic joints. Proteomic analysis confirmed that E4 strongly binds to macrophages and certain RA synovial fluid proteins such as α-enolase. The protective effect of E4 was epitope-specific and dependent on the interaction between E4-citrullinated α-enolase immune complexes with FCGR2B on macrophages, resulting in increased IL-10 secretion and reduced osteoclastogenesis. These findings suggest that a subset of ACPAs have therapeutic potential in RA.
© 2023. The Author(s).
Conflict of interest statement
C.G., B.X., T.R., and R.H. are the co-inventors of a patent application (PCT/EP2018/082236) related to ACPA specificity and their use in the diagnosis, prevention, and treatment of autoimmune diseases. H.U.S. and R.E.M.T. are mentioned as inventors on a patent on ACPA-IgG V-domain glycosylation. The remaining authors declare no competing interests.
Figures

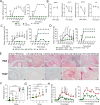




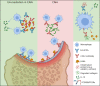
Comment in
-
A protective ACPA.Nat Immunol. 2023 Mar;24(3):377. doi: 10.1038/s41590-023-01458-5. Nat Immunol. 2023. PMID: 36829072 No abstract available.
-
Can ACPA protect against RA?Nat Rev Rheumatol. 2023 May;19(5):257. doi: 10.1038/s41584-023-00962-0. Nat Rev Rheumatol. 2023. PMID: 37020107 No abstract available.
References
-
- Heliövaara M, Aho K, Aromaa A, Knekt P, Reunanen A. Smoking and risk of rheumatoid arthritis. J. Rheumatol. 1993;20:1830–1835. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

