Genetic models of cleavage-reduced and soluble TREM2 reveal distinct effects on myelination and microglia function in the cuprizone model
- PMID: 36755323
- PMCID: PMC9909920
- DOI: 10.1186/s12974-022-02671-z
Genetic models of cleavage-reduced and soluble TREM2 reveal distinct effects on myelination and microglia function in the cuprizone model
Abstract
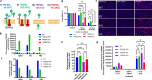
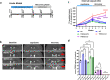
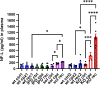
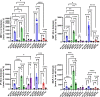
Triggering receptor expressed on myeloid cells 2 (TREM2) is a cell-surface immunoreceptor expressed on microglia, osteoclasts, dendritic cells and macrophages. Heterozygous loss-of-function mutations in TREM2, including mutations enhancing shedding form the cell surface, have been associated with myelin/neuronal loss and neuroinflammation in neurodegenerative diseases, such as Alzheimer`s disease and Frontotemporal Dementia. Using the cuprizone model, we investigated the involvement of soluble and cleavage-reduced TREM2 on central myelination processes in cleavage-reduced (TREM2-IPD), soluble-only (TREM2-sol), knockout (TREM2-KO) and wild-type (WT) mice. The TREM2-sol mouse is a new model with selective elimination of plasma membrane TREM2 and a reduced expression of soluble TREM2. In the acute cuprizone model demyelination and remyelination events were reflected by a T2-weighted signal intensity change in magnetic resonance imaging (MRI), most prominently in the external capsule (EC). In contrast to WT and TREM2-IPD, TREM2-sol and TREM2-KO showed an additional increase in MRI signal during the recovery phase. Histological analyses of TREM2-IPD animals revealed no recovery of neuroinflammation as well as of the lysosomal marker LAMP-1 and displayed enhanced cytokine/chemokine levels in the brain. TREM2-sol and, to a much lesser extent, TREM2-KO, however, despite presenting reduced levels of some cytokines/chemokines, showed persistent microgliosis and astrocytosis during recovery, with both homeostatic (TMEM119) as well as activated (LAMP-1) microglia markers increased. This was accompanied, specifically in the EC, by no myelin recovery, with appearance of myelin debris and axonal pathology, while oligodendrocytes recovered. In the chronic model consisting of 12-week cuprizone administration followed by 3-week recovery TREM2-IPD displayed sustained microgliosis and enhanced remyelination in the recovery phase. Taken together, our data suggest that sustained microglia activation led to increased remyelination, whereas microglia without plasma membrane TREM2 and only soluble TREM2 had reduced phagocytic activity despite efficient lysosomal function, as observed in bone marrow-derived macrophages, leading to a dysfunctional phenotype with improper myelin debris removal, lack of remyelination and axonal pathology following cuprizone intoxication.
Keywords: Cuprizone; MRI; Microglia; Neuroinflammation; Phagocytosis; TREM2.
© 2023. The Author(s).
Conflict of interest statement
All authors are or have been employees and shareholders of Novartis Pharma AG, Basel Switzerland.
Figures



Similar articles
-
Brain region-specific enhancement of remyelination and prevention of demyelination by the CSF1R kinase inhibitor BLZ945.Acta Neuropathol Commun. 2018 Feb 15;6(1):9. doi: 10.1186/s40478-018-0510-8. Acta Neuropathol Commun. 2018. PMID: 29448957 Free PMC article.
-
Acute TREM2 inhibition depletes MAFB-high microglia and hinders remyelination.Proc Natl Acad Sci U S A. 2025 Apr;122(13):e2426786122. doi: 10.1073/pnas.2426786122. Epub 2025 Mar 25. Proc Natl Acad Sci U S A. 2025. PMID: 40131948
-
TREM2 activation on microglia promotes myelin debris clearance and remyelination in a model of multiple sclerosis.Acta Neuropathol. 2020 Oct;140(4):513-534. doi: 10.1007/s00401-020-02193-z. Epub 2020 Aug 9. Acta Neuropathol. 2020. PMID: 32772264 Free PMC article.
-
The roles of microglia and astrocytes in phagocytosis and myelination: Insights from the cuprizone model of multiple sclerosis.Glia. 2022 Jul;70(7):1215-1250. doi: 10.1002/glia.24148. Epub 2022 Feb 2. Glia. 2022. PMID: 35107839 Free PMC article. Review.
-
The role of TREM2 in myelin sheath dynamics: A comprehensive perspective from physiology to pathology.Prog Neurobiol. 2025 Apr;247:102732. doi: 10.1016/j.pneurobio.2025.102732. Epub 2025 Feb 26. Prog Neurobiol. 2025. PMID: 40021075 Review.
Cited by
-
Magnetic resonance imaging and ultrasound elastography in the context of preclinical pharmacological research: significance for the 3R principles.Front Pharmacol. 2023 Jun 28;14:1177421. doi: 10.3389/fphar.2023.1177421. eCollection 2023. Front Pharmacol. 2023. PMID: 37448960 Free PMC article. Review.
-
Cholesterol in the CNS: functions, recycling and remyelination.J Neuroinflammation. 2025 Jul 11;22(1):180. doi: 10.1186/s12974-025-03490-8. J Neuroinflammation. 2025. PMID: 40646563 Free PMC article. Review.
-
Phospholipase D Family Member 4 Regulates Microglial Phagocytosis and Remyelination via the AKT Pathway in a Cuprizone-Induced Multiple Sclerosis Mouse Model.CNS Neurosci Ther. 2024 Nov;30(11):e70111. doi: 10.1111/cns.70111. CNS Neurosci Ther. 2024. PMID: 39548665 Free PMC article.
-
Berberine-loaded iron oxide nanoparticles alleviate cuprizone-induced astrocytic reactivity in a rat model of multiple sclerosis.Biometals. 2025 Feb;38(1):203-229. doi: 10.1007/s10534-024-00648-4. Epub 2024 Nov 14. Biometals. 2025. PMID: 39543075 Free PMC article.
-
Trem2-deficiency aggravates and accelerates age-related myelin degeneration.Acta Neuropathol Commun. 2024 Sep 19;12(1):154. doi: 10.1186/s40478-024-01855-3. Acta Neuropathol Commun. 2024. PMID: 39300502 Free PMC article.
References
-
- Bazenet C, Lovestone S. Plasma biomarkers for Alzheimer’s disease: much needed but tough to find. Biomark Med. 2012;6:441–454. - PubMed
-
- Feuerbach D, Schindler P, Barske C, Joller S, Beng-Louka E, Worringer KA, et al. ADAM17 is the main sheddase for the generation of human triggering receptor expressed in myeloid cells (hTREM2) ectodomain and cleaves TREM2 after Histidine 157. Neurosci Lett. 2017;660:109–114. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

