Osteogenic and anti-inflammatory effect of the multifunctional bionic hydrogel scaffold loaded with aspirin and nano-hydroxyapatite
- PMID: 36761294
- PMCID: PMC9902883
- DOI: 10.3389/fbioe.2023.1105248
Osteogenic and anti-inflammatory effect of the multifunctional bionic hydrogel scaffold loaded with aspirin and nano-hydroxyapatite
Erratum in
-
Corrigendum: Osteogenic and anti-inflammatory effect of the multifunctional bionic hydrogel scaffold loaded with aspirin and nano-hydroxyapatite.Front Bioeng Biotechnol. 2023 Mar 30;11:1179873. doi: 10.3389/fbioe.2023.1179873. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 37064221 Free PMC article.
Abstract
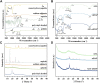
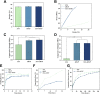
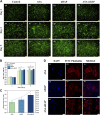
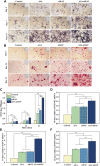
Although tissue engineering offered new approaches to repair bone defects, it remains a great challenge to create a bone-friendly microenvironment and rebuild bone tissue rapidly by a scaffold with a bionic structure. In this study, a multifunctional structurally optimized hydrogel scaffold was designed by integrating polyvinyl alcohol (PVA), gelatin (Gel), and sodium alginate (SA) with aspirin (ASA) and nano-hydroxyapatite (nHAP). The fabrication procedure is through a dual-crosslinking process. The chemical constitution, crystal structure, microstructure, porosity, mechanical strength, swelling and degradation property, and drug-release behavior of the hydrogel scaffold were analyzed. Multi-hydrogen bonds, electrostatic interactions, and strong "egg-shell" structure contributed to the multi-network microstructure, bone tissue-matched properties, and desirable drug-release function of the hydrogel scaffold. The excellent performance in improving cell viability, promoting cell osteogenic differentiation, and regulating the inflammatory microenvironment of the prepared hydrogel scaffold was verified using mouse pre-osteoblasts (MC3T3-E1) cells. And the synergistic osteogenic and anti-inflammatory functions of aspirin and nano-hydroxyapatite were also verified. This study provided valuable insights into the design, fabrication, and biological potential of multifunctional bone tissue engineering materials with the premise of constructing a bone-friendly microenvironment.
Keywords: aspirin; multifunctional hydrogel scaffold; nano-hydroxyapatite; sustained release; tissue engineering.
Copyright © 2023 Li, Xiaowen, Yang, Liu, Sun, Liu, Yin and Chen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Corrigendum: Osteogenic and anti-inflammatory effect of the multifunctional bionic hydrogel scaffold loaded with aspirin and nano-hydroxyapatite.Front Bioeng Biotechnol. 2023 Mar 30;11:1179873. doi: 10.3389/fbioe.2023.1179873. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 37064221 Free PMC article.
-
Fabrication and characterization of novel nano-biocomposite scaffold of chitosan-gelatin-alginate-hydroxyapatite for bone tissue engineering.Mater Sci Eng C Mater Biol Appl. 2016 Jul 1;64:416-427. doi: 10.1016/j.msec.2016.03.060. Epub 2016 Mar 22. Mater Sci Eng C Mater Biol Appl. 2016. PMID: 27127072
-
Sericin/Nano-Hydroxyapatite Hydrogels Based on Graphene Oxide for Effective Bone Regeneration via Immunomodulation and Osteoinduction.Int J Nanomedicine. 2023 Apr 6;18:1875-1895. doi: 10.2147/IJN.S399487. eCollection 2023. Int J Nanomedicine. 2023. PMID: 37051313 Free PMC article.
-
Bioactive injectable triple acting thermosensitive hydrogel enriched with nano-hydroxyapatite for bone regeneration: in-vitro characterization, Saos-2 cell line cell viability and osteogenic markers evaluation.Drug Dev Ind Pharm. 2019 May;45(5):787-804. doi: 10.1080/03639045.2019.1572184. Epub 2019 Feb 4. Drug Dev Ind Pharm. 2019. Retraction in: Drug Dev Ind Pharm. 2022 Dec;48(12):746. doi: 10.1080/03639045.2023.2165803. PMID: 30672348 Retracted.
-
Therapeutic Potential of Nano-Sustained-Release Factors for Bone Scaffolds.J Funct Biomater. 2025 Apr 9;16(4):136. doi: 10.3390/jfb16040136. J Funct Biomater. 2025. PMID: 40278244 Free PMC article. Review.
Cited by
-
Polysaccharide Hydroxyapatite (Nano)composites and Their Biomedical Applications: An Overview of Recent Years.ACS Omega. 2024 Jul 2;9(28):30035-30070. doi: 10.1021/acsomega.4c02170. eCollection 2024 Jul 16. ACS Omega. 2024. PMID: 39035931 Free PMC article. Review.
-
Osteogenic potential of silver nanoparticles in critical sized mandibular bone defects: an experimental study in white albino rats.Odontology. 2025 Jul;113(3):1062-1072. doi: 10.1007/s10266-024-01049-2. Epub 2025 Jan 10. Odontology. 2025. PMID: 39792286 Free PMC article.
-
Efficacy of Bone Morphogenetic Protein-2 Peptide-Modified Nano-Hydroxyapatite Alginate Hydrogel in Vertebral Bone Defect Repair.J Craniofac Surg. 2025 Jun 1;36(4):1226-1232. doi: 10.1097/SCS.0000000000010996. Epub 2025 Feb 25. J Craniofac Surg. 2025. PMID: 39998867 Free PMC article.
-
Characterization and biocompatibility of a bilayer PEEK-based scaffold for guiding bone regeneration.BMC Oral Health. 2024 Sep 27;24(1):1138. doi: 10.1186/s12903-024-04909-z. BMC Oral Health. 2024. PMID: 39334225 Free PMC article.
-
Advances in biomaterials for osteonecrosis treatment.Front Pharmacol. 2025 May 21;16:1559810. doi: 10.3389/fphar.2025.1559810. eCollection 2025. Front Pharmacol. 2025. PMID: 40469973 Free PMC article. Review.
References
-
- Bobbert F. S. L., Lietaert K., Eftekhari A. A., Pouran B., Ahmadi S. M., Weinans H., et al. (2017). Additively manufactured metallic porous biomaterials based on minimal surfaces: A unique combination of topological, mechanical, and mass transport properties. Acta Biomater. 53, 572–584. 10.1016/j.actbio.2017.02.024 - DOI - PubMed
-
- Cavagni J., Muniz F. W. M. G., Rösing C. K. (2016). The effect of inflammatory response modulator agents on gingivitis and periodontitis. RGO, Rev. Gaúch. Odontol. 64, 312–319. 10.1590/1981-8637201600030000112165 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

