1,4-Dithianes: attractive C2-building blocks for the synthesis of complex molecular architectures
- PMID: 36761474
- PMCID: PMC9907017
- DOI: 10.3762/bjoc.19.12
1,4-Dithianes: attractive C2-building blocks for the synthesis of complex molecular architectures
Abstract
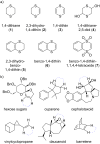
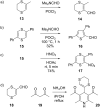
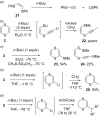
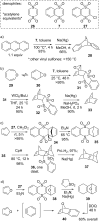
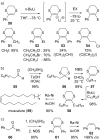
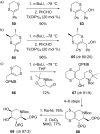
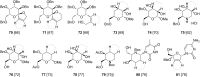
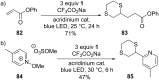
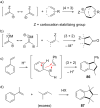
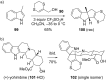
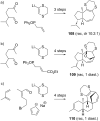
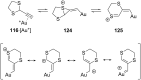
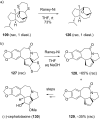
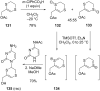
This review covers the synthetic applications of 1,4-dithianes, as well as derivatives thereof at various oxidation states. The selected examples show how the specific heterocyclic reactivity can be harnessed for the controlled synthesis of carbon-carbon bonds. The reactivity is compared to and put into context with more common synthetic building blocks, such as 1,3-dithianes and (hetero)aromatic building blocks. 1,4-Dithianes have as yet not been investigated to the same extent as their well-known 1,3-dithiane counterparts, but they do offer attractive transformations that can find good use in the assembly of a wide array of complex molecular architectures, ranging from lipids and carbohydrates to various carbocyclic scaffolds. This versatility arises from the possibility to chemoselectively cleave or reduce the sulfur-heterocycle to reveal a versatile C2-synthon.
Keywords: 1,4-dithianes; 1,4-dithiins; 2,3-dihydro-1,4-dithiins; heterocycles; target synthesis.
Copyright © 2023, Ryckaert et al.
Figures


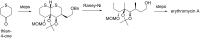







References
-
- Corey E J, Seebach D. Angew Chem, Int Ed Engl. 1965;4:1075–1077. doi: 10.1002/anie.196510752. - DOI
-
- Seebach D, Corey E J. J Org Chem. 1975;40:231–237. doi: 10.1021/jo00890a018. - DOI
-
- Seebach D. Angew Chem, Int Ed Engl. 1979;18:239–258. doi: 10.1002/anie.197902393. - DOI
-
- Yus M, Nájera C, Foubelo F. Tetrahedron. 2003;59:6147–6212. doi: 10.1016/s0040-4020(03)00955-4. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous
