Targeting NRF2 to promote epithelial repair
- PMID: 36762597
- PMCID: PMC9987932
- DOI: 10.1042/BST20220228
Targeting NRF2 to promote epithelial repair
Abstract
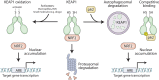
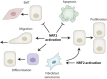
The transcription factor NRF2 is well known as a master regulator of the cellular stress response. As such, activation of NRF2 has gained widespread attention for its potential to prevent tissue injury, but also as a possible therapeutic approach to promote repair processes. While NRF2 activation affects most or even all cell types, its effect on epithelial cells during repair processes has been particularly well studied. In response to tissue injury, these cells proliferate, migrate and/or spread to effectively repair the damage. In this review, we discuss how NRF2 governs repair of epithelial tissues, and we highlight the increasing number of NRF2 targets with diverse roles in regulating epithelial repair.
Keywords: Keap1; NRF2; cell proliferation; cytoprotection; tissue repair; wound healing.
© 2023 The Author(s).
Conflict of interest statement
The authors declare that there are no competing interests associated with the manuscript.
Figures
Similar articles
-
Targeting the KEAP1-NRF2 System to Prevent Kidney Disease Progression.Am J Nephrol. 2017;45(6):473-483. doi: 10.1159/000475890. Epub 2017 May 13. Am J Nephrol. 2017. PMID: 28502971 Review.
-
Dysregulation of Nrf2/Keap1 Redox Pathway in Diabetes Affects Multipotency of Stromal Cells.Diabetes. 2019 Jan;68(1):141-155. doi: 10.2337/db18-0232. Epub 2018 Oct 23. Diabetes. 2019. PMID: 30352880 Free PMC article.
-
p62/SQSTM1 protects against cisplatin-induced oxidative stress in kidneys by mediating the cross talk between autophagy and the Keap1-Nrf2 signalling pathway.Free Radic Res. 2019 Jul;53(7):800-814. doi: 10.1080/10715762.2019.1635251. Epub 2019 Jul 8. Free Radic Res. 2019. PMID: 31223046
-
Overexpression of miR-200a protects cardiomyocytes against hypoxia-induced apoptosis by modulating the kelch-like ECH-associated protein 1-nuclear factor erythroid 2-related factor 2 signaling axis.Int J Mol Med. 2016 Oct;38(4):1303-11. doi: 10.3892/ijmm.2016.2719. Epub 2016 Aug 26. Int J Mol Med. 2016. PMID: 27573160
-
Stress-sensing mechanisms and the physiological roles of the Keap1-Nrf2 system during cellular stress.J Biol Chem. 2017 Oct 13;292(41):16817-16824. doi: 10.1074/jbc.R117.800169. Epub 2017 Aug 24. J Biol Chem. 2017. PMID: 28842501 Free PMC article. Review.
Cited by
-
Targeting Signalling Pathways in Chronic Wound Healing.Int J Mol Sci. 2023 Dec 19;25(1):50. doi: 10.3390/ijms25010050. Int J Mol Sci. 2023. PMID: 38203220 Free PMC article. Review.
-
Mechanism of Nrf2 in the treatment of ulcerative colitis via regulating macrophage polarization.Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2023 Nov 28;48(11):1746-1752. doi: 10.11817/j.issn.1672-7347.2023.230281. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2023. PMID: 38432866 Free PMC article. Review. Chinese, English.
-
Screening and Antioxidant Activities Evaluation of Peptides From Abalone (Haliotis discus hannai Ino).Food Sci Nutr. 2025 Feb 9;13(2):e70028. doi: 10.1002/fsn3.70028. eCollection 2025 Feb. Food Sci Nutr. 2025. PMID: 39926104 Free PMC article.
-
Platelet-Released Growth Factors (PRGFs) Activate NRF2-ARE and Modulate Inflammatory Response in an NRF2-Dependent Manner in Primary Human Keratinocytes.J Cosmet Dermatol. 2025 May;24(5):e70228. doi: 10.1111/jocd.70228. J Cosmet Dermatol. 2025. PMID: 40353553 Free PMC article.
-
The ferroptotic effect of NRF2-GCLM signaling axis derived by radiotherapy of esophageal squamous cell cancer: the vivo study.Sci Rep. 2025 Jul 24;15(1):26917. doi: 10.1038/s41598-025-10414-2. Sci Rep. 2025. PMID: 40707557 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

