Co-dependent regulation of p-BRAF and potassium channel KCNMA1 levels drives glioma progression
- PMID: 36763212
- PMCID: PMC9918570
- DOI: 10.1007/s00018-023-04708-9
Co-dependent regulation of p-BRAF and potassium channel KCNMA1 levels drives glioma progression
Abstract
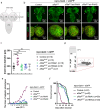
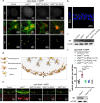
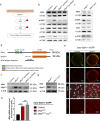
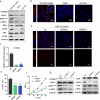
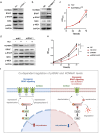
BRAF mutations have been found in gliomas which exhibit abnormal electrophysiological activities, implying their potential links with the ion channel functions. In this study, we identified the Drosophila potassium channel, Slowpoke (Slo), the ortholog of human KCNMA1, as a critical factor involved in dRafGOF glioma progression. Slo was upregulated in dRafGOF glioma. Knockdown of slo led to decreases in dRafGOF levels, glioma cell proliferation, and tumor-related phenotypes. Overexpression of slo in glial cells elevated dRaf expression and promoted cell proliferation. Similar mutual regulations of p-BRAF and KCNMA1 levels were then recapitulated in human glioma cells with the BRAF mutation. Elevated p-BRAF and KCNMA1 were also observed in HEK293T cells upon the treatment of 20 mM KCl, which causes membrane depolarization. Knockdown KCNMA1 in these cells led to a further decrease in cell viability. Based on these results, we conclude that the levels of p-BRAF and KCNMA1 are co-dependent and mutually regulated. We propose that, in depolarized glioma cells with BRAF mutations, high KCNMA1 levels act to repolarize membrane potential and facilitate cell growth. Our study provides a new strategy to antagonize the progression of gliomas as induced by BRAF mutations.
Keywords: BRAF; Glioma; KCNMA1; Potassium channel.
© 2023. This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply.
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose.
Figures


Similar articles
-
Comparative gain-of-function effects of the KCNMA1-N999S mutation on human BK channel properties.J Neurophysiol. 2020 Feb 1;123(2):560-570. doi: 10.1152/jn.00626.2019. Epub 2019 Dec 18. J Neurophysiol. 2020. PMID: 31851553 Free PMC article.
-
An emerging spectrum of variants and clinical features in KCNMA1-linked channelopathy.Channels (Austin). 2021 Dec;15(1):447-464. doi: 10.1080/19336950.2021.1938852. Channels (Austin). 2021. PMID: 34224328 Free PMC article. Review.
-
Further studies of ion channels in the electroreceptor of the skate through deep sequencing, cloning and cross species comparisons.Gene. 2019 Nov 15;718:143989. doi: 10.1016/j.gene.2019.143989. Epub 2019 Jul 18. Gene. 2019. PMID: 31326551
-
Conserved Role of the Large Conductance Calcium-Activated Potassium Channel, KCa1.1, in Sinus Node Function and Arrhythmia Risk.Circ Genom Precis Med. 2021 Apr;14(2):e003144. doi: 10.1161/CIRCGEN.120.003144. Epub 2021 Feb 25. Circ Genom Precis Med. 2021. PMID: 33629867 Free PMC article.
-
KCNMA1-linked channelopathy.J Gen Physiol. 2019 Oct 7;151(10):1173-1189. doi: 10.1085/jgp.201912457. Epub 2019 Aug 19. J Gen Physiol. 2019. PMID: 31427379 Free PMC article. Review.
Cited by
-
The BK Channel Limits the Pro-Inflammatory Activity of Macrophages.Cells. 2024 Feb 9;13(4):322. doi: 10.3390/cells13040322. Cells. 2024. PMID: 38391935 Free PMC article.
-
Transcriptomics reveals age-related changes in ion transport-related factors in yak lungs.Front Vet Sci. 2024 May 8;11:1374794. doi: 10.3389/fvets.2024.1374794. eCollection 2024. Front Vet Sci. 2024. PMID: 38779034 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

