Connected Droplet Shape Analysis for Nanoflow Quantification in Thin Electroosmotic Micropumps and a Tunable Convex Lens Application
- PMID: 36763988
- PMCID: PMC9949215
- DOI: 10.1021/acs.langmuir.2c02875
Connected Droplet Shape Analysis for Nanoflow Quantification in Thin Electroosmotic Micropumps and a Tunable Convex Lens Application
Abstract
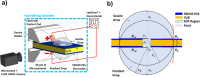
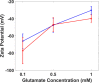
Thin electroosmotic flow (EOF) micropumps can generate flow in confined spaces such as lab-on-a-chip microsystems and implantable drug delivery devices. However, status quo methods for quantifying flow and other important parameters in EOF micropumps depend on microfluidic interconnects or fluorescent particle tracking: methods that can be complex and error-prone. Here, we present a novel connected droplet shape analysis (CDSA) technique that simplifies flow rate and zeta potential quantification in thin EOF micropumps. We also show that a pair of droplets connected by an EOF pump can function as a tunable convex lens system (TCLS). We developed a biocompatible and all polymer EOF micropump with an SU-8 substrate and poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) electrodes. We microdrilled a channel through the electrode/SU-8/electrode layers to realize a monolithic EOF micropump. Then, we deposited a pinned droplet on each end of the microchannel so that it connected them. By controlling the EOF between the droplets and measuring the corresponding change in their shape, we quantified the nanoliter EOF rate and zeta potential at the interface of SU-8 with two liquids (deionized water and a l-glutamate neurotransmitter solution). When the droplet pair and pump were used as a TCLS, CDSA successfully predicted how the focal length would change when the pump drove fluid from one droplet to another. In summary, CDSA is a simple low-cost technique for EOF rate and zeta potential measurement, and a pair of droplets connected by an EOF micropump can function as a TCLS without any moving parts.
Conflict of interest statement
The authors declare no competing financial interest.
Figures






Similar articles
-
Microfluidic flow control on charged phospholipid polymer interface.Lab Chip. 2007 Feb;7(2):199-206. doi: 10.1039/b616851p. Epub 2006 Dec 5. Lab Chip. 2007. PMID: 17268622
-
A liquid metal based, integrated parallel electroosmotic micropump cluster drive system.Lab Chip. 2024 Feb 13;24(4):896-903. doi: 10.1039/d3lc00926b. Lab Chip. 2024. PMID: 38263786
-
Control of particle-deposition pattern in a sessile droplet by using radial electroosmotic flow.Anal Chem. 2006 Jul 15;78(14):5192-7. doi: 10.1021/ac0601866. Anal Chem. 2006. PMID: 16841947
-
The Electroosmotic Flow (EOF).Methods Mol Biol. 2010;583:121-34. doi: 10.1007/978-1-60327-106-6_5. Methods Mol Biol. 2010. PMID: 19763462 Review.
-
From Ion Current to Electroosmotic Flow Rectification in Asymmetric Nanopore Membranes.Nanomaterials (Basel). 2017 Dec 14;7(12):445. doi: 10.3390/nano7120445. Nanomaterials (Basel). 2017. PMID: 29240676 Free PMC article. Review.
Cited by
-
A Miniaturized Archimedean Screw Pump for High-Viscosity Fluid Pumping in Microfluidics.Micromachines (Basel). 2023 Jul 12;14(7):1409. doi: 10.3390/mi14071409. Micromachines (Basel). 2023. PMID: 37512720 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

