Non-Conventional Allogeneic Anti-BCMA Chimeric Antigen Receptor-Based Immune Cell Therapies for Multiple Myeloma Treatment
- PMID: 36765526
- PMCID: PMC9913487
- DOI: 10.3390/cancers15030567
Non-Conventional Allogeneic Anti-BCMA Chimeric Antigen Receptor-Based Immune Cell Therapies for Multiple Myeloma Treatment
Abstract
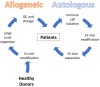
MM, characterized by the progressive accumulation of clonal plasma cells in bone marrow, remains a severe medical problem globally. Currently, almost all MM patients who have received standard treatments will eventually relapse. Autologous anti-BCMA CAR-T cells are one of the FDA-approved immunotherapy cell-based products for treating adults with relapsed or refractory (r/r) multiple myeloma. However, this type of CAR-T cell product has several limitations, including high costs, long manufacturing times, and possible manufacturing failure, which significantly hinder its wider application for more patients. In this review, we summarized the current development stage of applying other types of immune cells to bring the anti-BCMA CAR-T therapy from autologous to allogeneic. In general, anti-BCMA CAR gene-edited αβ T cells and CAR-Natural Killer (NK) cells are at the forefront, with multiple clinical trials ongoing, while CAR-γδ T cells and CAR-invariant Natural Killer T (iNKT) cells are still in pre-clinical studies. Other immune cells such as macrophages, B cells, and dendritic cells have been mainly developed to target other antigens and have the potential to be used to target BCMA. Nevertheless, additional regulatory requirements might need to be taken into account in developing these non-conventional allogenic anti-BCMA CAR-based cell products.
Keywords: anti-BCMA CAR; clinical regulations; immune cells; multiple myeloma; non-conventional; off-the-shelf.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; the collection, analysis, or interpretation of data; the writing of the manuscript; or the decision to publish the results.
Figures
Similar articles
-
Anti-myeloma efficacy of CAR-iNKT is enhanced with a long-acting IL-7, rhIL-7-hyFc.Blood Adv. 2023 Oct 24;7(20):6009-6022. doi: 10.1182/bloodadvances.2023010032. Blood Adv. 2023. PMID: 37399471 Free PMC article.
-
Preclinical Evaluation of Allogeneic CAR T Cells Targeting BCMA for the Treatment of Multiple Myeloma.Mol Ther. 2019 Jun 5;27(6):1126-1138. doi: 10.1016/j.ymthe.2019.04.001. Epub 2019 Apr 8. Mol Ther. 2019. PMID: 31005597 Free PMC article.
-
Overview of anti-BCMA CAR-T immunotherapy for multiple myeloma and relapsed/refractory multiple myeloma.Scand J Immunol. 2020 Aug;92(2):e12910. doi: 10.1111/sji.12910. Epub 2020 Jun 17. Scand J Immunol. 2020. PMID: 32471019 Review.
-
Allogeneic Anti-BCMA CAR T Cells Are Superior to Multiple Myeloma-derived CAR T Cells in Preclinical Studies and May Be Combined with Gamma Secretase Inhibitors.Cancer Res Commun. 2022 Mar 23;2(3):158-171. doi: 10.1158/2767-9764.CRC-21-0157. eCollection 2022 Mar. Cancer Res Commun. 2022. PMID: 36874402 Free PMC article.
-
Ciltacabtagene autoleucel: The second anti-BCMA CAR T-cell therapeutic armamentarium of relapsed or refractory multiple myeloma.Front Immunol. 2022 Sep 2;13:991092. doi: 10.3389/fimmu.2022.991092. eCollection 2022. Front Immunol. 2022. PMID: 36119032 Free PMC article. Review.
Cited by
-
Contribution of long-lived plasma cells to antibody-mediated allograft rejection.Clin Transplant Res. 2024 Dec 31;38(4):341-353. doi: 10.4285/ctr.24.0047. Epub 2024 Dec 18. Clin Transplant Res. 2024. PMID: 39690904 Free PMC article. Review.
-
Advancing Multiple Myeloma Immunotherapy: A Review of Chimeric Antigen Receptor T-Cell and Bispecific T-Cell Engagers Cell Therapies in Revolutionizing Treatment.Iran J Med Sci. 2025 Jan 1;50(1):1-10. doi: 10.30476/ijms.2024.101739.3446. eCollection 2025 Jan. Iran J Med Sci. 2025. PMID: 39957814 Free PMC article. Review.
-
T Cell Based Immunotherapy for Cancer: Approaches and Strategies.Vaccines (Basel). 2023 Apr 13;11(4):835. doi: 10.3390/vaccines11040835. Vaccines (Basel). 2023. PMID: 37112747 Free PMC article. Review.
-
Translating B cell immunology to the treatment of antibody-mediated allograft rejection.Nat Rev Nephrol. 2024 Apr;20(4):218-232. doi: 10.1038/s41581-023-00791-0. Epub 2024 Jan 2. Nat Rev Nephrol. 2024. PMID: 38168662 Review.
-
Updates on CAR T cell therapy in multiple myeloma.Biomark Res. 2024 Sep 12;12(1):102. doi: 10.1186/s40364-024-00634-5. Biomark Res. 2024. PMID: 39261906 Free PMC article. Review.
References
-
- Cowan A.J., Allen C., Barac A., Basaleem H., Bensenor I., Curado M.P., Foreman K., Gupta R., Harvey J., Hosgood H.D., et al. Global Burden of Multiple Myeloma: A Systematic Analysis for the Global Burden of Disease Study 2016. JAMA Oncol. 2018;4:1221–1227. doi: 10.1001/jamaoncol.2018.2128. - DOI - PMC - PubMed
-
- Key Statistics about Multiple Myeloma. [(accessed on 28 September 2022)]. Available online: https://www.cancer.org/cancer/multiple-myeloma/about/key-statistics.html.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials