Legumes Regulate Symbiosis with Rhizobia via Their Innate Immune System
- PMID: 36769110
- PMCID: PMC9917363
- DOI: 10.3390/ijms24032800
Legumes Regulate Symbiosis with Rhizobia via Their Innate Immune System
Abstract
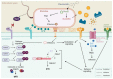
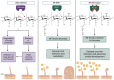
Plant roots are constantly exposed to a diverse microbiota of pathogens and mutualistic partners. The host's immune system is an essential component for its survival, enabling it to monitor nearby microbes for potential threats and respond with a defence response when required. Current research suggests that the plant immune system has also been employed in the legume-rhizobia symbiosis as a means of monitoring different rhizobia strains and that successful rhizobia have evolved to overcome this system to infect the roots and initiate nodulation. With clear implications for host-specificity, the immune system has the potential to be an important target for engineering versatile crops for effective nodulation in the field. However, current knowledge of the interacting components governing this pathway is limited, and further research is required to build on what is currently known to improve our understanding. This review provides a general overview of the plant immune system's role in nodulation. With a focus on the cycles of microbe-associated molecular pattern-triggered immunity (MTI) and effector-triggered immunity (ETI), we highlight key molecular players and recent findings while addressing the current knowledge gaps in this area.
Keywords: legumes; nodulation; pathogen; plant immune system; rhizobia; symbiosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Gresshoff P.M., Hayashi S., Biswas B., Mirzaei S., Indrasumunar A., Reid D., Samuel S., Tollenaere A., van Hameren B., Hastwell A., et al. The value of biodiversity in legume symbiotic nitrogen fixation and nodulation for biofuel and food production. J. Plant Physiol. 2015;172:128–136. doi: 10.1016/j.jplph.2014.05.013. - DOI - PubMed
-
- Jensen E.S., Peoples M.B., Boddey R.M., Gresshoff P.M., Hauggaard-Nielsen H., Alves B.J.R., Morrison M.J. Legumes for mitigation of climate change and the provision of feedstock for biofuels and biorefineries. A review. Agron. Sustain. Dev. 2012;32:329–364. doi: 10.1007/s13593-011-0056-7. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

