Wilms Tumor 1-Driven Fibroblast Activation and Subpleural Thickening in Idiopathic Pulmonary Fibrosis
- PMID: 36769178
- PMCID: PMC9918078
- DOI: 10.3390/ijms24032850
Wilms Tumor 1-Driven Fibroblast Activation and Subpleural Thickening in Idiopathic Pulmonary Fibrosis
Abstract
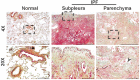
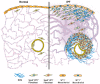
Idiopathic pulmonary fibrosis (IPF) is a progressive fibrotic lung disease that is often fatal due to the formation of irreversible scar tissue in the distal areas of the lung. Although the pathological and radiological features of IPF lungs are well defined, the lack of insight into the fibrogenic role of fibroblasts that accumulate in distinct anatomical regions of the lungs is a critical knowledge gap. Fibrotic lesions have been shown to originate in the subpleural areas and extend into the lung parenchyma through processes of dysregulated fibroproliferation, migration, fibroblast-to-myofibroblast transformation, and extracellular matrix production. Identifying the molecular targets underlying subpleural thickening at the early and late stages of fibrosis could facilitate the development of new therapies to attenuate fibroblast activation and improve the survival of patients with IPF. Here, we discuss the key cellular and molecular events that contribute to (myo)fibroblast activation and subpleural thickening in IPF. In particular, we highlight the transcriptional programs involved in mesothelial to mesenchymal transformation and fibroblast dysfunction that can be targeted to alter the course of the progressive expansion of fibrotic lesions in the distal areas of IPF lungs.
Keywords: collagen; extracellular matrix; fibroblast; idiopathic pulmonary fibrosis; lung function.
Conflict of interest statement
The authors declare that they have no conflict of interest to report regarding the present study.
Figures
Similar articles
-
Wilms' tumor 1 drives fibroproliferation and myofibroblast transformation in severe fibrotic lung disease.JCI Insight. 2018 Aug 23;3(16):e121252. doi: 10.1172/jci.insight.121252. eCollection 2018 Aug 23. JCI Insight. 2018. PMID: 30135315 Free PMC article.
-
Wilms tumor 1 impairs apoptotic clearance of fibroblasts in distal fibrotic lung lesions.J Clin Invest. 2025 Jun 10;135(15):e188819. doi: 10.1172/JCI188819. eCollection 2025 Aug 1. J Clin Invest. 2025. PMID: 40493404 Free PMC article.
-
Fibrocytes Regulate Wilms Tumor 1-Positive Cell Accumulation in Severe Fibrotic Lung Disease.J Immunol. 2015 Oct 15;195(8):3978-91. doi: 10.4049/jimmunol.1500963. Epub 2015 Sep 14. J Immunol. 2015. PMID: 26371248 Free PMC article.
-
New therapeutics based on emerging concepts in pulmonary fibrosis.Expert Opin Ther Targets. 2019 Jan;23(1):69-81. doi: 10.1080/14728222.2019.1552262. Epub 2018 Nov 28. Expert Opin Ther Targets. 2019. PMID: 30468628 Free PMC article. Review.
-
Time for a change: is idiopathic pulmonary fibrosis still idiopathic and only fibrotic?Lancet Respir Med. 2018 Feb;6(2):154-160. doi: 10.1016/S2213-2600(18)30007-9. Lancet Respir Med. 2018. PMID: 29413083 Free PMC article. Review.
Cited by
-
Nε-Carboxymethyl-Lysine Modification of Extracellular Matrix Proteins Augments Fibroblast Activation.Int J Mol Sci. 2023 Oct 31;24(21):15811. doi: 10.3390/ijms242115811. Int J Mol Sci. 2023. PMID: 37958795 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

