Recent Developments in Carbon-11 Chemistry and Applications for First-In-Human PET Studies
- PMID: 36770596
- PMCID: PMC9920299
- DOI: 10.3390/molecules28030931
Recent Developments in Carbon-11 Chemistry and Applications for First-In-Human PET Studies
Abstract
Positron emission tomography (PET) is a molecular imaging technique that makes use of radiolabelled molecules for in vivo evaluation. Carbon-11 is a frequently used radionuclide for the labelling of small molecule PET tracers and can be incorporated into organic molecules without changing their physicochemical properties. While the short half-life of carbon-11 (11C; t½ = 20.4 min) offers other advantages for imaging including multiple PET scans in the same subject on the same day, its use is limited to facilities that have an on-site cyclotron, and the radiochemical transformations are consequently more restrictive. Many researchers have embraced this challenge by discovering novel carbon-11 radiolabelling methodologies to broaden the synthetic versatility of this radionuclide. This review presents new carbon-11 building blocks and radiochemical transformations as well as PET tracers that have advanced to first-in-human studies over the past five years.
Keywords: Carbon-11; first-in-human; positron emission tomography (PET); radiochemistry; radiotracer.
Conflict of interest statement
The authors declare no relevant conflict of interest.
Figures
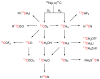




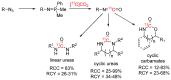
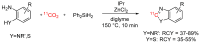

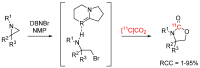

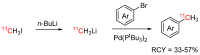





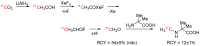


References
-
- Bernard-Gauthier V., Lepage M.L., Waengler B., Bailey J.J., Liang S.H., Perrin D.M., Vasdev N., Schirrmacher R. Recent Advances in 18F Radiochemistry: A Focus on B-18F, Si-18F, Al-18F, and C-18F Radiofluorination via Spirocyclic Iodonium Ylides. J. Nucl. Med. 2018;59:568–572. doi: 10.2967/jnumed.117.197095. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

