Antimicrobial Natural Products from Plant Pathogenic Fungi
- PMID: 36770808
- PMCID: PMC9920077
- DOI: 10.3390/molecules28031142
Antimicrobial Natural Products from Plant Pathogenic Fungi
Abstract
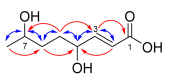
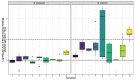
Isolates of a variety of fungal plant pathogens (Alternaria radicina ICMP 5619, Cercospora beticola ICMP 15907, Dactylonectria macrodidyma ICMP 16789, D. torresensis ICMP 20542, Ilyonectria europaea ICMP 16794, and I. liriodendra ICMP 16795) were screened for antimicrobial activity against the human pathogenic bacteria Acinetobacter baumannii, Pseudomonas aeruginosa, Escherichia coli, Mycobacterium abscessus, and M. marinum and were found to have some activity. Investigation of the secondary metabolites of these fungal isolates led to the isolation of ten natural products (1-10) of which one was novel, (E)-4,7-dihydroxyoct-2-enoic acid (1). Structure elucidation of all natural products was achieved by a combination of NMR spectroscopy and mass spectrometry. We also investigated the antimicrobial activity of a number of the isolated natural products. While we did not find (E)-4,7-dihydroxyoct-2-enoic acid (1) to have any activity against the bacteria and fungi in our assays, we did find that cercosporin (7) exhibited potent activity against Methicillin resistant Staphylococcus aureus (MRSA), dehydro-curvularin (6) and radicicol (10) exhibited antimycobacterial activity against M. marinum, and brefeldin A (8) and radicicol (10) exhibited antifungal activity against Candida albicans. Investigation of the cytotoxicity and haemolytic activities of these natural products (6-8 and 10) found that only one of the four active compounds, radicicol (10), was non-cytotoxic and non-haemolytic.
Keywords: Candida albicans; Staphylocccus aureus; antibacterial activity; fungal plant pathogens; isolation; metabolites; mycobacteria; natural product.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

