Exploring the Co-Crystallization of Kojic Acid with Silver(I), Copper(II), Zinc(II), and Gallium(III) for Potential Antibacterial Applications
- PMID: 36770910
- PMCID: PMC9920434
- DOI: 10.3390/molecules28031244
Exploring the Co-Crystallization of Kojic Acid with Silver(I), Copper(II), Zinc(II), and Gallium(III) for Potential Antibacterial Applications
Abstract
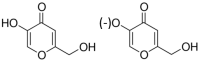
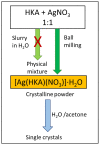
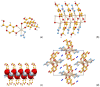
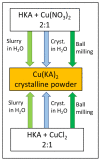
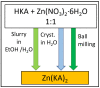
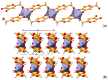
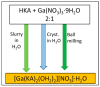
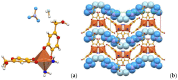
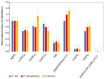
Co-crystallization of kojic acid (HKA) with silver(I), copper(II), zinc(II), or gallium(III) salts yielded three 1D coordination polymers and one 0D complex in which kojic acid was present as a neutral or anionic terminal or bridging ligand. All reactions were conducted mechanochemically via ball milling and manual grinding, or via slurry. All solids were fully characterized via single-crystal and/or powder X-ray diffraction. As kojic acid is a mild antimicrobial compound that is widely used in cosmetics, and the metal cations possess antibacterial properties, their combinations were tested for potential antibacterial applications. The minimal inhibition concentrations (MICs) and minimal biocidal concentrations (MBCs) for all compounds were measured against standard strains of the bacteria P. aeruginosa, S. aureus, and E. coli. All compounds exerted appreciable antimicrobial activity in the order of silver, zinc, copper, and gallium complexes.
Keywords: antimicrobial metals; antimicrobials; co-crystallization; copper; gallium; mechanochemistry; silver; zinc.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- 2019 Antibacterial Agents in Clinical Development: An Analysis of the Antibacterial Clinical Development Pipeline. World Health Organization; Geneva, Switzerland: 2019.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

