The Functional Roles of ISG15/ISGylation in Cancer
- PMID: 36771004
- PMCID: PMC9918931
- DOI: 10.3390/molecules28031337
The Functional Roles of ISG15/ISGylation in Cancer
Abstract
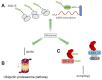
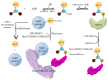
The protein ISG15 encoded by interferon-stimulated gene (ISG) 15 is the first identified member of the ubiquitin-like protein family and exists in the form of monomers and conjugated complexes. Like ubiquitin, ISG15 can mediate an ubiquitin-like modification by covalently modifying other proteins, known as ISGylation. There is growing evidence showing that both the free and conjugated ISG15 are involved in multiple key cellular processes, including autophagy, exosome secretion, DNA repair, immune regulation, and cancer occurrence and progression. In this review, we aim to further clarify the function of ISG15 and ISGylation in cancer, demonstrate the important relationship between ISG15/ISGylation and cancer, and emphasize new insights into the different roles of ISG15/ISGylation in cancer progression. This review may contribute to therapeutic intervention in cancer. However, due to the limitations of current research, the regulation of ISG15/ISGylation on cancer progression is not completely clear, thus further comprehensive and sufficient correlation studies are still needed.
Keywords: ISG15; ISGylation; cancer; post-translational modification; ubiquitin-like protein.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
ISG15 in cancer: Beyond ubiquitin-like protein.Cancer Lett. 2018 Dec 1;438:52-62. doi: 10.1016/j.canlet.2018.09.007. Epub 2018 Sep 11. Cancer Lett. 2018. PMID: 30213559 Review.
-
Interferon-stimulated gene 15 and the protein ISGylation system.J Interferon Cytokine Res. 2011 Jan;31(1):119-30. doi: 10.1089/jir.2010.0110. Epub 2010 Dec 29. J Interferon Cytokine Res. 2011. PMID: 21190487 Free PMC article. Review.
-
The diverse repertoire of ISG15: more intricate than initially thought.Exp Mol Med. 2022 Nov;54(11):1779-1792. doi: 10.1038/s12276-022-00872-3. Epub 2022 Nov 1. Exp Mol Med. 2022. PMID: 36319753 Free PMC article. Review.
-
ISG15 and ISGylation in Human Diseases.Cells. 2022 Feb 4;11(3):538. doi: 10.3390/cells11030538. Cells. 2022. PMID: 35159348 Free PMC article. Review.
-
Curcumin partly prevents ISG15 activation via ubiquitin-activating enzyme E1-like protein and decreases ISGylation.Biochem Biophys Res Commun. 2022 Oct 15;625:94-101. doi: 10.1016/j.bbrc.2022.08.003. Epub 2022 Aug 5. Biochem Biophys Res Commun. 2022. PMID: 35952613 Free PMC article.
Cited by
-
Unveiling the Multifaceted Roles of ISG15: From Immunomodulation to Therapeutic Frontiers.Vaccines (Basel). 2024 Feb 1;12(2):153. doi: 10.3390/vaccines12020153. Vaccines (Basel). 2024. PMID: 38400136 Free PMC article. Review.
-
A single-cell atlas characterizes dysregulation of the bone marrow immune microenvironment associated with outcomes in multiple myeloma.bioRxiv [Preprint]. 2024 May 17:2024.05.15.593193. doi: 10.1101/2024.05.15.593193. bioRxiv. 2024. PMID: 38798338 Free PMC article. Preprint.
-
Epigenetic modification of m6A regulator proteins in cancer.Mol Cancer. 2023 Jun 30;22(1):102. doi: 10.1186/s12943-023-01810-1. Mol Cancer. 2023. PMID: 37391814 Free PMC article. Review.
-
ISG15 promotes tumor progression via IL6/JAK2/STAT3 signaling pathway in ccRCC.Clin Exp Med. 2024 Jun 29;24(1):140. doi: 10.1007/s10238-024-01414-z. Clin Exp Med. 2024. PMID: 38951255 Free PMC article.
-
Evolution of the tumor immune landscape during treatment with tebentafusp, a T cell receptor-CD3 bispecific.Cell Rep Med. 2025 Apr 15;6(4):102076. doi: 10.1016/j.xcrm.2025.102076. Cell Rep Med. 2025. PMID: 40239619 Free PMC article. Clinical Trial.
References
-
- Schmid S., Hugel T., Institute of Physical Chemistry. University of Freiburg. Germany. Signalling research centers BIOSS. Cibss. Albert Ludwigs University Germany Controlling protein function by fine-tuning conformational flexibility. Elife. 2020;9:e57180. doi: 10.7554/eLife.57180. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

