Amyloid-like RIP1/RIP3 RHIM Fragments' Characterization and Application as a Drug Depot
- PMID: 36771145
- PMCID: PMC9918910
- DOI: 10.3390/molecules28031480
Amyloid-like RIP1/RIP3 RHIM Fragments' Characterization and Application as a Drug Depot
Abstract
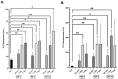
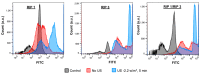
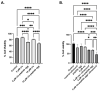
Amyloid aggregates play a major role in diseases as well as in normal physiological function. Receptor-interacting protein kinases 1 and 3 (RIP1/RIP3) aggregates complexes in cellular necroptosis is one example of protein aggregation in normal cellular function. Although recently there have been several studies on full kinase proteins aggregation, the aggregation potential of small peptide sequences of RIP1/RIP3, the physicochemical properties, and the potential in biomedical applications have not been explored. Hence, in this paper, we study the aggregation propensity of peptides consisting of four and twelve amino acid sequences in the RHIM region of RIP1/RIP3 proteins that are known to drive the beta-sheet formation and the subsequent aggregation. The aggregation kinetics, physicochemical characterization, mechanosensitive properties, cellular effects, and potential as a cancer drug depot have been investigated. The results show that the number and concentration of amino acids play a role in amyloid-like aggregates' properties. Further, the aggregates when formulated with cisplatin-induced significant lung cancer cell toxicity compared to an equal amount of cisplatin with and without ultrasound. The study would serve as a platform for further investigation on RIP1/RIP3 peptide and protein aggregates, their role in multiple cellular functions and diseases, and their potential as drug depots.
Keywords: Amyloid; RIP1; RIP3; aggregation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
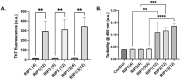



References
-
- Hafner Bratkovic I. Prions, prionoid complexes and amyloids: The bad, the good and something in between. Swiss Med. Wkly. 2017;147:w14424. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

