Protocatechuic Acid Prevents Some of the Memory-Related Behavioural and Neurotransmitter Changes in a Pyrithiamine-Induced Thiamine Deficiency Model of Wernicke-Korsakoff Syndrome in Rats
- PMID: 36771332
- PMCID: PMC9921060
- DOI: 10.3390/nu15030625
Protocatechuic Acid Prevents Some of the Memory-Related Behavioural and Neurotransmitter Changes in a Pyrithiamine-Induced Thiamine Deficiency Model of Wernicke-Korsakoff Syndrome in Rats
Abstract
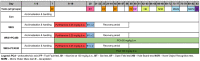
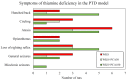
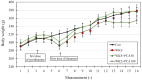
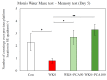
The purpose of this research was to investigate the effects of protocatechuic acid (PCA) at doses of 50 and 100 mg/kg on the development of unfavourable changes in cognitive processes in a pyrithiamine-induced thiamine deficiency (PTD) model of the Wernicke-Korsakoff syndrome (WKS) in rats. The effects of PCA were assessed at the behavioural and biochemical levels. Behavioural analysis was conducted using the Foot Fault test (FF), Bar test, Open Field test, Novel Object Recognition test (NOR), Hole-Board test and Morris Water Maze test (MWM). Biochemical analysis consisting of determination of concentration and turnover of neurotransmitters in selected structures of the rat CNS was carried out using high-performance liquid chromatography. PTD caused catalepsy (Bar test) and significantly impaired motor functions, leading to increased ladder crossing time and multiplied errors due to foot misplacement (FF). Rats with experimentally induced WKS showed impaired consolidation and recall of spatial reference memory in the MWM test, while episodic memory related to object recognition in the NOR was unimpaired. Compared to the control group, rats with WKS showed reduced serotonin levels in the prefrontal cortex and changes in dopamine and/or norepinephrine metabolites in the prefrontal cortex, medulla oblongata and spinal cord. PTD was also found to affect alanine, serine, glutamate, and threonine levels in certain areas of the rat brain. PCA alleviated PTD-induced cataleptic symptoms in rats, also improving their performance in the Foot Fault test. In the MWM, PCA at 50 and 100 mg/kg b.w. improved memory consolidation and the ability to retrieve acquired information in rats, thereby preventing unfavourable changes caused by PTD. PCA at both tested doses was also shown to have a beneficial effect on normalising PTD-disrupted alanine and glutamate concentrations in the medulla oblongata. These findings demonstrate that certain cognitive deficits in spatial memory and abnormalities in neurotransmitter levels persist in rats that have experienced an acute episode of PTD, despite restoration of thiamine supply and long-term recovery. PCA supplementation largely had a preventive effect on the development of these deficits, to some extent also normalising neurotransmitter concentrations in the brain.
Keywords: Wernicke–Korsakoff syndrome; behaviour; glutamate; memory; neurotransmitters; protocatechuic acid.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






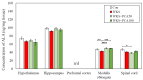
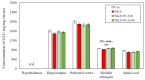
References
-
- Kimura A., Sugimoto T., Kitamori K., Saji N., Niida S., Toba K., Sakurai T. Malnutrition is associated with behavioral and psychiatric symptoms of dementia in older women with mild cognitive impairment and early-stage Alzheimer’s disease. Nutrients. 2019;11:1951. doi: 10.3390/nu11081951. - DOI - PMC - PubMed
-
- Widy-Tyszkiewicz E. Current Evidence for Disease Prevention and Treatment by Protocatechuic Acid (PCA) and Its Precursor Protocatechuic Aldehyde (PCAL) in Animals and Humans. In: Ekiert H.M., Ramawat K.G., Arora J., editors. Plant Antioxidants and Health. Springer; Berlin/Heidelberg, Germany: 2022. (Reference Series in Phytochemistry). - DOI
-
- Krzysztoforska K., Mirowska-Guzel D., Widy-Tyszkiewicz E. Pharmacological effects of protocatechuic acid and its therapeutic potential in neurodegenerative diseases: Review on the basis of in vitro and in vivo studies in rodents and humans. Nutr. Neurosci. 2017;22:72–82. doi: 10.1080/1028415X.2017.1354543. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

