Ring-Opening Polymerization of Trimethylene Carbonate with Phosphazene Organocatalyst
- PMID: 36772021
- PMCID: PMC9921643
- DOI: 10.3390/polym15030720
Ring-Opening Polymerization of Trimethylene Carbonate with Phosphazene Organocatalyst
Abstract
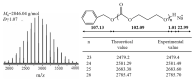
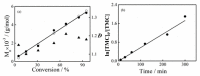
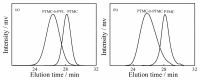
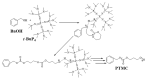
Aliphatic polycarbonate (APC) compounds are an important class of biodegradable materials with excellent biocompatibility, good biodegradability, and low toxicity, and the study of these compounds and their modification products aims to obtain biodegradable materials with better performance. In this context, the ring-opening polymerization (ROP) of trimethylene carbonate (TMC) from a low nucleophilic organic superbase of phosphazene (t-BuP4) as a catalyst and benzyl alcohol (BnOH) as an initiator at room temperature was carefully studied to prepare poly(trimethylene carbonate) (PTMC) which is one of the most studied APC. 1H NMR and SEC measurements clearly demonstrate the presence of a benzyloxy group at the α-terminus of the obtained PTMC homopolymers while investigation of the polymerization kinetics confirms the controlled/living nature of t-BuP4-catalyzed ROP of TMC. On the basis of this, the block copolymerization of TMC and δ-valerolactone (VL)/ε-caprolactone (CL) was successfully carried out to give PTMC-b-PCL and PTMC-b-PVL copolymers. Furthermore, PTMC with terminal functionality was also prepared with the organocatalytic ROP of TMC through functional initiators. We believe that the present ROP system is a robust, highly efficient, and practical strategy for producing excellent biocompatible and biodegradable PTMC-based materials.
Keywords: PTMC; controlled/living nature; end functionality; t-BuP4.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

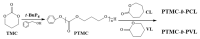



References
-
- Dirauf M., Muljajew I., Weber C., Schubert U.S. Recent Advances in Degradable Synthetic Polymers for Biomedical Applications-Beyond Polyesters. Prog. Polym. Sci. 2022;129:101547. doi: 10.1016/j.progpolymsci.2022.101547. - DOI
-
- Saghebasl S., Akbarzadeh A., Gorabi A.M., Nikzamir N., SeyedSadjadi M., Mostafavi E. Biodegradable Functional Macromolecules as Promising Scaffolds for Cardiac Tissue Engineering. Polym. Adv. Technol. 2022;33:2044–2068. doi: 10.1002/pat.5669. - DOI
-
- Fukushima K., Fukushima K. Biodegradable Functional Biomaterials Exploiting Substituted Trimethylene Carbonates and Organocatalytic Transesterification. Polym. J. 2016;48:1103–1114. doi: 10.1038/pj.2016.80. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

