The Parkinson-Associated Toxin Paraquat Shifts Physiological α-Synuclein Tetramers toward Monomers That Can Be Calpain-Truncated and Form Oligomers
- PMID: 36773784
- PMCID: PMC10155269
- DOI: 10.1016/j.ajpath.2023.01.010
The Parkinson-Associated Toxin Paraquat Shifts Physiological α-Synuclein Tetramers toward Monomers That Can Be Calpain-Truncated and Form Oligomers
Abstract
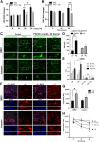
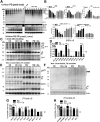
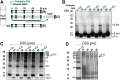
Abnormal aggregation of α-synuclein (αS) is thought to initiate neuronal dysfunction and death in Parkinson disease (PD). In addition to higher-molecular-weight, oligomeric, and polymeric forms of αS associated with neurotoxicity and disease, recent findings indicate the occurrence of physiological tetrameric assemblies in healthy neurons in culture and in brain. Herein, the PD-associated neurotoxin paraquat reduced physiological tetramers and led to calpain-truncated monomers and an approximately 70-kDa apparent oligomer different in size from physiological αS multimers. These truncated and oligomeric forms could also be generated by calpain cleavage of pure, recombinant human αS in vitro. Moreover, they were detected in the brains of tetramer-abrogating, E46K-amplified (3K) mice that model PD. These results indicate that paraquat triggers membrane damage and aberrant calpain activity that can induce a pathologic shift of tetramers toward an excess of full-length and truncated monomers, the accumulation of αS oligomers, and insoluble cytoplasmic αS puncta. The findings suggest that an environmental precipitant of PD can alter αS tetramer/monomer equilibrium, as already shown for several genetically caused forms of PD.
Copyright © 2023 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Female Sex and Brain-Selective Estrogen Benefit α-Synuclein Tetramerization and the PD-like Motor Syndrome in 3K Transgenic Mice.J Neurosci. 2019 Sep 18;39(38):7628-7640. doi: 10.1523/JNEUROSCI.0313-19.2019. Epub 2019 Aug 12. J Neurosci. 2019. PMID: 31405930 Free PMC article.
-
Parkinson-causing α-synuclein missense mutations shift native tetramers to monomers as a mechanism for disease initiation.Nat Commun. 2015 Jun 16;6:7314. doi: 10.1038/ncomms8314. Nat Commun. 2015. PMID: 26076669 Free PMC article.
-
KTKEGV repeat motifs are key mediators of normal α-synuclein tetramerization: Their mutation causes excess monomers and neurotoxicity.Proc Natl Acad Sci U S A. 2015 Aug 4;112(31):9596-601. doi: 10.1073/pnas.1505953112. Epub 2015 Jul 7. Proc Natl Acad Sci U S A. 2015. PMID: 26153422 Free PMC article.
-
Defining the native state of α-synuclein.Neurodegener Dis. 2014;13(2-3):114-7. doi: 10.1159/000355516. Epub 2013 Oct 30. Neurodegener Dis. 2014. PMID: 24192542 Review.
-
Interactions of α-synuclein oligomers with lipid membranes.Biochim Biophys Acta Biomembr. 2021 Apr 1;1863(4):183536. doi: 10.1016/j.bbamem.2020.183536. Epub 2020 Dec 26. Biochim Biophys Acta Biomembr. 2021. PMID: 33373595 Review.
Cited by
-
LRP10 and α-synuclein transmission in Lewy body diseases.Cell Mol Life Sci. 2024 Feb 5;81(1):75. doi: 10.1007/s00018-024-05135-0. Cell Mol Life Sci. 2024. PMID: 38315424 Free PMC article.
-
Synucleinopathies: Intrinsic and Extrinsic Factors.Cell Biochem Biophys. 2023 Sep;81(3):427-442. doi: 10.1007/s12013-023-01154-z. Epub 2023 Aug 1. Cell Biochem Biophys. 2023. PMID: 37526884
-
Parkinson disease-associated toxic exposures selectively up-regulate vesicular glutamate transporter vGlut2 in a model of human cortical neurons.Mol Biol Cell. 2025 Feb 1;36(2):br4. doi: 10.1091/mbc.E24-08-0376. Epub 2025 Jan 2. Mol Biol Cell. 2025. PMID: 39745872 Free PMC article.
-
Research Priorities on the Role of α-Synuclein in Parkinson's Disease Pathogenesis.Mov Disord. 2024 Oct;39(10):1663-1678. doi: 10.1002/mds.29897. Epub 2024 Jun 30. Mov Disord. 2024. PMID: 38946200 Free PMC article. Review.
-
Aggregation-resistant alpha-synuclein tetramers are reduced in the blood of Parkinson's patients.EMBO Mol Med. 2024 Jul;16(7):1657-1674. doi: 10.1038/s44321-024-00083-5. Epub 2024 Jun 5. EMBO Mol Med. 2024. PMID: 38839930 Free PMC article.
References
-
- Koprich J.B., Kalia L.V., Brotchie J.M. Animal models of alpha-synucleinopathy for Parkinson disease drug development. Nat Rev Neurosci. 2017;18:515–529. - PubMed
-
- El-Gamal M., Salama M., Collins-Praino L.E., Baetu I., Fathalla A.M., Soliman A.M., Mohamed W., Moustafa A.A. Neurotoxin-induced rodent models of Parkinson's disease: benefits and drawbacks. Neurotox Res. 2021;39:897–923. - PubMed
-
- Nuber S., Tadros D., Fields J., Overk C.R., Ettle B., Kosberg K., Mante M., Rockenstein E., Trejo M., Masliah E. Environmental neurotoxic challenge of conditional alpha-synuclein transgenic mice predicts a dopaminergic olfactory-striatal interplay in early PD. Acta Neuropathol. 2014;127:477–494. - PMC - PubMed
-
- Schlachetzki J.C., Grimm T., Schlachetzki Z., Ben Abdallah N.M., Ettle B., Vohringer P., Ferger B., Winner B., Nuber S., Winkler J. Dopaminergic lesioning impairs adult hippocampal neurogenesis by distinct modification of alpha-synuclein. J Neurosci Res. 2016;94:62–73. - PubMed
-
- Wang W., Nguyen L.T., Burlak C., Chegini F., Guo F., Chataway T., Ju S., Fisher O.S., Miller D.W., Datta D., Wu F., Wu C.X., Landeru A., Wells J.A., Cookson M.R., Boxer M.B., Thomas C.J., Gai W.P., Ringe D., Petsko G.A., Hoang Q.Q. Caspase-1 causes truncation and aggregation of the Parkinson's disease-associated protein alpha-synuclein. Proc Natl Acad Sci U S A. 2016;113:9587–9592. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

