Humoral and cellular immune correlates of protection against COVID-19 in kidney transplant recipients
- PMID: 36773936
- PMCID: PMC9911984
- DOI: 10.1016/j.ajt.2023.02.015
Humoral and cellular immune correlates of protection against COVID-19 in kidney transplant recipients
Abstract
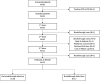
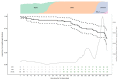
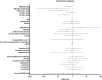
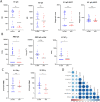
As solid organ transplant recipients are at high risk of severe COVID-19 and respond poorly to primary SARS-CoV-2 mRNA vaccination, they have been prioritized for booster vaccination. However, an immunological correlate of protection has not been identified in this vulnerable population. We conducted a prospective monocentric cohort study of 65 kidney transplant recipients who received 3 doses of BNT162b2 mRNA vaccine. Associations among breakthrough infection (BTI), vaccine responses, and patient characteristics were explored in 54 patients. Symptomatic COVID-19 was diagnosed in 32% of kidney transplant recipients during a period of 6 months after booster vaccination. During this period, SARS-CoV-2 delta and omicron were the dominant variants in the general population. Univariate Analyses identified the avidity of SARS-CoV-2 receptor binding domain binding IgG, neutralizing antibodies, and SARS-CoV-2 S2-specific interferon gamma responses as correlates of protection against BTI. No demographic or clinical parameter correlated with the risk of BTI. In multivariate analysis, the risk of BTI was best predicted by neutralizing antibody and S2-specific interferon gamma responses. In conclusion, T cell responses may help compensate for the suboptimal antibody response to booster vaccination in kidney transplant recipients. Further studies are needed to confirm these findings.
Keywords: COVID-19; breakthrough cases; immunogenicity; kidney transplantation; mRNA vaccination.
Copyright © 2023 American Society of Transplantation & American Society of Transplant Surgeons. Published by Elsevier Inc. All rights reserved.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

