Neuroprotective Effects of a Multi-Herbal Extract on Axonal and Synaptic Disruption in Vitro and Cognitive Impairment in Vivo
- PMID: 36777330
- PMCID: PMC9912829
- DOI: 10.3233/ADR-220056
Neuroprotective Effects of a Multi-Herbal Extract on Axonal and Synaptic Disruption in Vitro and Cognitive Impairment in Vivo
Abstract
Background: Alzheimer's disease (AD) is a multifactorial disorder characterized by cognitive decline. Current available therapeutics for AD have limited clinical benefit. Therefore, preventive therapies for interrupting the development of AD are critically needed. Molecules targeting multifunction to interact with various pathlogical components have been considered to improve the therapeutic efficiency of AD. In particular, herbal medicines with multiplicity of actions produce cognitive benefits on AD. Bugu-M is a multi-herbal extract composed of Ganoderma lucidum (Antler form), Nelumbo nucifera Gaertn., Ziziphus jujuba Mill., and Dimocarpus longan, with the ability of its various components to confer resilience to cognitive deficits.
Objective: To evaluate the potential of Bugu-M on amyloid-β (Aβ) toxicity and its in vitro mechanisms and on in vivo cognitive function.
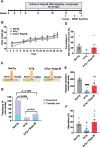
Methods: We illustrated the effect of Bugu-M on Aβ25-35-evoked toxicity as well as its possible mechanisms to diminish the pathogenesis of AD in rat cortical neurons. For cognitive function studies, 2-month-old female 3×Tg-AD mice were administered 400 mg/kg Bugu-M for 30 days. Behavioral tests were performed to assess the efficacy of Bugu-M on cognitive impairment.
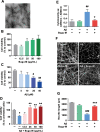
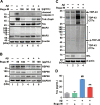
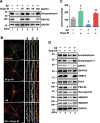
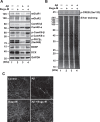
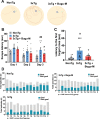
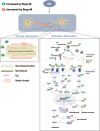
Results: In primary cortical neuronal cultures, Bugu-M mitigated Aβ-evoked toxicity by reducing cytoskeletal aberrations and axonal disruption, restoring presynaptic and postsynaptic protein expression, suppressing mitochondrial damage and apoptotic signaling, and reserving neurogenic and neurotrophic factors. Importantly, 30-day administration of Bugu-M effectively prevented development of cognitive impairment in 3-month-old female 3×Tg-AD mice.
Conclusion: Bugu-M might be beneficial in delaying the progression of AD, and thus warrants consideration for its preventive potential for AD.
Keywords: Alzheimer’s disease; amyloid-β; axon; cognition; dementia; herbal; mild cognitive impairment; natural product; prevention; synapse.
© 2023 – The authors. Published by IOS Press.
Conflict of interest statement
The authors have no conflict of interest to report.
Figures

Similar articles
-
Preventive Effects of Continuous Betaine Intake on Cognitive Impairment and Aberrant Gene Expression in Hippocampus of 3xTg Mouse Model of Alzheimer's Disease.J Alzheimers Dis. 2021;79(2):639-652. doi: 10.3233/JAD-200972. J Alzheimers Dis. 2021. PMID: 33337369
-
Tripchlorolide improves cognitive deficits by reducing amyloid β and upregulating synapse-related proteins in a transgenic model of Alzheimer's Disease.J Neurochem. 2015 Apr;133(1):38-52. doi: 10.1111/jnc.13056. Epub 2015 Mar 8. J Neurochem. 2015. PMID: 25661995
-
Prevention of Amyloid-β and Tau Pathologies, Associated Neurodegeneration, and Cognitive Deficit by Early Treatment with a Neurotrophic Compound.J Alzheimers Dis. 2017;58(1):215-230. doi: 10.3233/JAD-170075. J Alzheimers Dis. 2017. PMID: 28387677
-
Alzheimer's disease.Subcell Biochem. 2012;65:329-52. doi: 10.1007/978-94-007-5416-4_14. Subcell Biochem. 2012. PMID: 23225010 Review.
-
Literature review on the interdisciplinary biomarkers of multi-target and multi-time herbal medicine therapy to modulate peripheral systems in cognitive impairment.Front Neurosci. 2023 Feb 16;17:1108371. doi: 10.3389/fnins.2023.1108371. eCollection 2023. Front Neurosci. 2023. PMID: 36875644 Free PMC article. Review.
Cited by
-
Coupling of Alzheimer's Disease Genetic Risk Factors with Viral Susceptibility and Inflammation.Aging Dis. 2024 Oct 1;15(5):2028-2050. doi: 10.14336/AD.2023.1017. Aging Dis. 2024. PMID: 37962454 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

