Novel nutraceutical supplements with yeast β-glucan, prebiotics, minerals, and Silybum marianum (silymarin) ameliorate obesity-related metabolic and clinical parameters: A double-blind randomized trial
- PMID: 36778595
- PMCID: PMC9912840
- DOI: 10.3389/fendo.2022.1089938
Novel nutraceutical supplements with yeast β-glucan, prebiotics, minerals, and Silybum marianum (silymarin) ameliorate obesity-related metabolic and clinical parameters: A double-blind randomized trial
Abstract
Purpose: It is known that obesity has a multifactorial etiology that involves genetic and environmental factors. The WHO estimates the worldwide prevalence of 1.9 billion overweight adults and more than 650 million people with obesity. These alarming data highlight the high and growing prevalence of obesity and represent a risk factor for the development and aggravation of other chronic diseases, such as nonalcoholic fatty liver disease (NAFLD) that is frequently considered the hepatic outcome of type 2 diabetes. The use of non-pharmacological therapies such as food supplements, nutraceuticals, and natural integrative therapies has grown as an alternative tool for obesity-related diseases compared to conventional medications. However, it is a still little explored research field and lacks scientific evidence of therapeutic effectiveness. Considering this, the aim is to evaluate whether a new nutraceutical supplement composition can improve and supply essential mineral nutrients, providing an improvement of obesity-related metabolic and endocrine parameters.
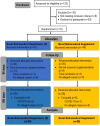
Methods: Sedentary volunteers (women and men) with body mass index (BMI) ≤34.9 kg/m2 were divided into two groups: Novel Nutraceutical Supplement_(S) (n = 30) and Novel Nutraceutical Supplement (n = 29), differing in the absence (S) or presence of silymarin, respectively. Volunteers were instructed to take two capsules in the morning and two capsules in the evening. No nutritional intervention was performed during the study period. The data (anthropometrics and anamneses) and harvest blood (biochemistry and hormonal exams) were collected at three different time points: baseline time [day 0 (T0)], day 90 (T90), and day 180 (T180) post-supplementation.
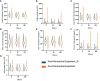
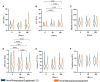
Results: In the anthropometric analysis, the waist circumference in middle abdomen (WC-mid) and waist circumference in iliac crest (WC-IC) were reduced. Also, the waist-to-height ratio (WHt R) and waist-to-hip ratio (WHR) seem to slightly decrease alongside the supplementation period with both nutraceutical supplements tested as well as transaminase enzyme ratio [aspartate aminotransferase (AST)/alanine aminotransferase (ALT) ratio (AAR)], a known as a biomarker of NAFLD, and endocrine hormones cortisol and thyroid-stimulating hormone (TSH) at 90 and 180 days post-supplementation.
Conclusions: In a condition associated with sedentary and no nutritional intervention, the new nutraceutical supplement composition demonstrated the ability to be a strong and newfangled tool to improve important biomarkers associated with obesity and its comorbidities.
Trial registration: ClinicalTrials.gov NCT04810572.
Keywords: Silybum marianum; endocrine parameters; nutraceutics; obesity; prebiotic; supplement; transaminases.
Copyright © 2023 Nehmi-Filho, Santamarina, de Freitas, Trarbach, de Oliveira, Palace-Berl, de Souza, de Miranda, Escamilla-Garcia, Otoch and Pessoa.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- WHO . The power of cities: tackling noncommunicable diseases and road safety. Geneva: World Health Organization; 2019 (WHO/NMH/PND/2019.9). Licence: CC BY-NC-SA 3.0 IGO.
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

