Metabolomic profiles of the liquid state fermentation in co-culture of Eurotium amstelodami and Bacillus licheniformis
- PMID: 36778878
- PMCID: PMC9909110
- DOI: 10.3389/fmicb.2023.1080743
Metabolomic profiles of the liquid state fermentation in co-culture of Eurotium amstelodami and Bacillus licheniformis
Abstract
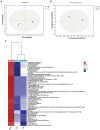
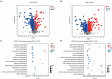
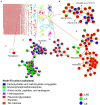
As an important source of new drug molecules, secondary metabolites (SMs) produced by microorganisms possess important biological activities, such as antibacterial, anti-inflammatory, and hypoglycemic effects. However, the true potential of microbial synthesis of SMs has not been fully elucidated as the SM gene clusters remain silent under laboratory culture conditions. Herein, we evaluated the inhibitory effect of Staphylococcus aureus by co-culture of Eurotium amstelodami and three Bacillus species, including Bacillus licheniformis, Bacillus subtilis, and Bacillus amyloliquefaciens. In addition, a non-target approach based on ultra-performance liquid chromatography time-of-flight mass spectrometry (UPLC-TOF-MS) was used to detect differences in extracellular and intracellular metabolites. Notably, the co-culture of E. amstelodami and Bacillus spices significantly improved the inhibitory effect against S. aureus, with the combination of E. amstelodami and B. licheniformis showing best performance. Metabolomics data further revealed that the abundant SMs, such as Nummularine B, Lucidenic acid E2, Elatoside G, Aspergillic acid, 4-Hydroxycyclohexylcarboxylic acid, Copaene, and Pipecolic acid were significantly enhanced in co-culture. Intracellularly, the differential metabolites were involved in the metabolism of amino acids, nucleic acids, and glycerophospholipid. Overall, this work demonstrates that the co-culture strategy is beneficial for inducing biosynthesis of active metabolites in E. amstelodami and B. licheniformis.
Keywords: Bacillus licheniformis; Eurotium amstelodami; co-culture; metabolomic profiles; secondary metabolism.
Copyright © 2023 Wang, Chen, Xin, Chen, Xu, He, Pan and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
UPLC-TOF/MS-based metabolomics reveals the chemical changes and in vitro biological effects in fermentation of white ginseng by four probiotics.Front Microbiol. 2022 Nov 24;13:1022200. doi: 10.3389/fmicb.2022.1022200. eCollection 2022. Front Microbiol. 2022. PMID: 36504795 Free PMC article.
-
Co-culture of Aspergillus sydowii and Bacillus subtilis induces the production of antibacterial metabolites.Fungal Biol. 2022 Apr;126(4):320-332. doi: 10.1016/j.funbio.2022.01.002. Epub 2022 Jan 31. Fungal Biol. 2022. PMID: 35314063
-
Protective Mechanism of Eurotium amstelodami from Fuzhuan Brick Tea against Colitis and Gut-Derived Liver Injury Induced by Dextran Sulfate Sodium in C57BL/6 Mice.Nutrients. 2024 Apr 16;16(8):1178. doi: 10.3390/nu16081178. Nutrients. 2024. PMID: 38674869 Free PMC article.
-
Exploring the Metabolomic Responses of Bacillus licheniformis to Temperature Stress by Gas Chromatography/Mass Spectrometry.J Microbiol Biotechnol. 2018 Mar 28;28(3):473-481. doi: 10.4014/jmb.1708.08019. J Microbiol Biotechnol. 2018. PMID: 29539884
-
Cryptic Chemical Communication: Secondary Metabolic Responses Revealed by Microbial Co-culture.Chem Asian J. 2020 Feb 3;15(3):327-337. doi: 10.1002/asia.201901505. Epub 2020 Jan 20. Chem Asian J. 2020. PMID: 31957936 Review.
Cited by
-
Chuanxiong Renshen Decoction Inhibits Alzheimer's Disease Neuroinflammation by Regulating PPARγ/NF-κB Pathway.Drug Des Devel Ther. 2024 Jul 24;18:3209-3232. doi: 10.2147/DDDT.S462266. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 39071817 Free PMC article.
-
Physicochemical properties and bioturbation analysis of high-temperature Daqu by functional microflora.Front Microbiol. 2025 Jun 26;16:1601675. doi: 10.3389/fmicb.2025.1601675. eCollection 2025. Front Microbiol. 2025. PMID: 40641880 Free PMC article.
-
Multi-omics profiling reveals the molecular mechanism of Bifidobacterium animalis BB04 in co-culture with Wickerhamomyces anomalus Y-5 to induce bifidocin A synthesis.World J Microbiol Biotechnol. 2024 Oct 26;40(11):366. doi: 10.1007/s11274-024-04172-y. World J Microbiol Biotechnol. 2024. PMID: 39455466
-
Co-Culture of White Rot Fungi Pleurotus ostreatus P5 and Bacillus amyloliquefaciens B2: A Strategy to Enhance Lipopeptide Production and Suppress of Fusarium Wilt of Cucumber.J Fungi (Basel). 2023 Oct 26;9(11):1049. doi: 10.3390/jof9111049. J Fungi (Basel). 2023. PMID: 37998854 Free PMC article.
-
Computation-aided studies related to the induction of specialized metabolite biosynthesis in microbial co-cultures: An introductory overview.Comput Struct Biotechnol J. 2023 Aug 16;21:4021-4029. doi: 10.1016/j.csbj.2023.08.011. eCollection 2023. Comput Struct Biotechnol J. 2023. PMID: 37649711 Free PMC article. Review.
References
-
- Arguelles-Arias A., Ongena M., Halimi B., Lara Y., Brans A., Joris B., et al. . (2009). Bacillus amyloliquefaciens GA1 as a source of potent antibiotics and other secondary metabolites for biocontrol of plant pathogens. Microb. Cell Factories 8:63. doi: 10.1186/1475-2859-8-63, PMID: - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

