Challenges, current status and emerging strategies in the development of rapidly dissolving FDM 3D-printed tablets: An overview and commentary
- PMID: 36778904
- PMCID: PMC9909727
- DOI: 10.5599/admet.1622
Challenges, current status and emerging strategies in the development of rapidly dissolving FDM 3D-printed tablets: An overview and commentary
Abstract
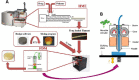
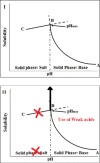
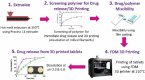
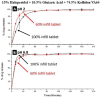
Since the approval of a 3D-printed tablet by the FDA in 2015 for marketing, there has been a great interest in 3D printing in the pharmaceutical field for the development of personalized and on-demand medications. Among various 3D printing methods explored for the development of oral solid dosage form like tablet, the fused deposition modeling (FDM) 3D-printing, where the drug-polymer mixtures are first converted into filaments by hot melt extrusion (HME) and then the filaments are printed into tablets using 3D printers by applying computer-aided design principles, has emerged as the most attractive option. However, no FDM 3D-printed tablets have yet been marketed as the technology faces many challenges, such as limited availability of pharmaceutical-grade polymers that can be printed into tablets, low drug-polymer miscibility, the need for high temperature for HME and 3D-printing, and slow drug release rates from tablets. These challenges are discussed in this article with a special focus on drug release rates since FDM 3D-printing usually leads to the preparation of slow-release tablets while the rapid release from dosage forms is often desired for optimal therapeutic outcomes of new drug candidates. Pros and cons of various strategies for the development of rapidly dissolving FDM 3D-printed tablets reported in the literature are reviewed. Finally, two case studies on emerging strategies for the development of rapidly dissolving FDM 3D-printed tablets are presented, where one outlines a systematic approach for formulating rapidly dissolving tablets, and the other describes a novel strategy to increase dissolution rates of drugs from FDM 3D-printed tablets, which at the same time can also increase drug-polymer miscibility and printability of tablets and lower processing temperatures. Thus, this overview and commentary discusses various issues involving the formulation of rapidly dissolving FDM 3D-printed tablets and provides guidance for the development of commercially viable products.
Keywords: 3D printing; 3D-printed tablet; drug release; fused deposition modeling; hot melt extrusion; rapid dissolution.
Copyright © 2023 by the authors.
Conflict of interest statement
Conflict of interest : The author declares no conflict of interest.
Figures




Similar articles
-
Improving drug release rate, drug-polymer miscibility, printability and processability of FDM 3D-printed tablets by weak acid-base interaction.Int J Pharm. 2023 Feb 5;632:122542. doi: 10.1016/j.ijpharm.2022.122542. Epub 2022 Dec 22. Int J Pharm. 2023. PMID: 36566823
-
A Lower Temperature FDM 3D Printing for the Manufacture of Patient-Specific Immediate Release Tablets.Pharm Res. 2016 Nov;33(11):2704-12. doi: 10.1007/s11095-016-1995-0. Epub 2016 Aug 9. Pharm Res. 2016. PMID: 27506424
-
Formulation of 3D Printed Tablet for Rapid Drug Release by Fused Deposition Modeling: Screening Polymers for Drug Release, Drug-Polymer Miscibility and Printability.J Pharm Sci. 2018 Jan;107(1):390-401. doi: 10.1016/j.xphs.2017.10.021. Epub 2017 Oct 21. J Pharm Sci. 2018. PMID: 29066279
-
A Review on Physicochemical Properties of Polymers Used as Filaments in 3D-Printed Tablets.AAPS PharmSciTech. 2023 May 9;24(5):116. doi: 10.1208/s12249-023-02570-3. AAPS PharmSciTech. 2023. PMID: 37160772 Review.
-
Polymer Selection for Hot-Melt Extrusion Coupled to Fused Deposition Modelling in Pharmaceutics.Pharmaceutics. 2020 Aug 22;12(9):795. doi: 10.3390/pharmaceutics12090795. Pharmaceutics. 2020. PMID: 32842703 Free PMC article. Review.
Cited by
-
Dissolution-permeation of hot-melt extruded amorphous solid dispersion comprising an experimental grade of HPMCAS.ADMET DMPK. 2023 Jul 22;11(3):373-385. doi: 10.5599/admet.1586. eCollection 2023. ADMET DMPK. 2023. PMID: 37829323 Free PMC article.
-
Status of Polymer Fused Deposition Modeling (FDM)-Based Three-Dimensional Printing (3DP) in the Pharmaceutical Industry.Polymers (Basel). 2024 Jan 30;16(3):386. doi: 10.3390/polym16030386. Polymers (Basel). 2024. PMID: 38337275 Free PMC article. Review.
-
Geometry-Driven Fabrication of Mini-Tablets via 3D Printing: Correlating Release Kinetics with Polyhedral Shapes.Pharmaceutics. 2024 Jun 8;16(6):783. doi: 10.3390/pharmaceutics16060783. Pharmaceutics. 2024. PMID: 38931904 Free PMC article.
-
The Influence of Polylactic Acid Filament Moisture Content on Dust Emissions in 3D Printing Process.Sensors (Basel). 2024 Dec 10;24(24):7890. doi: 10.3390/s24247890. Sensors (Basel). 2024. PMID: 39771629 Free PMC article.
References
-
- Awad A., Goyanes A., Basit A., Zidan A., Xu C., Li D., Narayan R., Chen R.K.. A Review of State-of-The-Art on Enabling Additive Manufacturing Processes for Precision Medicine. Journal of Manufacturing Science and Engineering 145 (2023) 010802. https://doi.org/10.1115/1.4056199 10.1115/1.4056199 - DOI
-
- Wong K.V., Hernandez A.. A review of additive manufacturing. International scholarly research notices 2012 (2012) 208760. https://doi.org/10.5402/2012/208760 10.5402/2012/208760 - DOI
-
- Spritam FDA approval history. https://www.drugs.com/history/spritam.html (last accessed: November 12, 2022)
-
- Melocchi A., Uboldi M., Cerea M., Foppoli A., Maroni A., Moutaharrik S., Palugan L., Zema L., Gazzaniga A.. A graphical review on the escalation of fused deposition modeling (FDM) 3D printing in the pharmaceutical field. Journal of Pharmaceutical Sciences 109(10) (2020) 2943-2957. https://doi.org/10.1016/j.xphs.2020.07.011 10.1016/j.xphs.2020.07.011 - DOI - PubMed
-
- Wang J., Zhang Y., Aghda N.H., Pillai A.R., Thakkar R., Nokhodchi A., Maniruzzaman M.. Emerging 3D printing technologies for drug delivery devices: Current status and future perspective. Advanced Drug Delivery Reviews 174 (2021) 294-316. https://doi.org/10.1016/j.addr.2021.04.019 10.1016/j.addr.2021.04.019 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous
