The gasdermin protein family: emerging roles in gastrointestinal health and disease
- PMID: 36781958
- PMCID: PMC10238632
- DOI: 10.1038/s41575-023-00743-w
The gasdermin protein family: emerging roles in gastrointestinal health and disease
Abstract
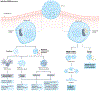
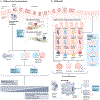
Since the identification and characterization of gasdermin (GSDM) D as the main effector of inflammatory regulated cell death (or pyroptosis), literature on the GSDM family of pore-forming proteins is rapidly expanding, revealing novel mechanisms regulating their expression and functions that go beyond pyroptosis. Indeed, a growing body of evidence corroborates the importance of GSDMs within the gastrointestinal system, underscoring their critical contributions to the pathophysiology of gastrointestinal cancers, enteric infections and gut mucosal inflammation, such as inflammatory bowel disease. However, with this increase in knowledge, several important and controversial issues have arisen regarding basic GSDM biology and its role(s) during health and disease states. These include critical questions centred around GSDM-dependent lytic versus non-lytic functions, the biological activities of cleaved versus full-length proteins, the differential roles of GSDM-expressing mucosal immune versus epithelial cells, and whether GSDMs promote pathogenic or protective effects during specific disease settings. This Review provides a comprehensive summary and interpretation of the current literature on GSDM biology, specifically focusing on the gastrointestinal tract, highlighting the main controversial issues and their clinical implications, and addressing future areas of research to unravel the specific role(s) of this intriguing, yet enigmatic, family of proteins.
© 2023. Springer Nature Limited.
Figures

References
-
- Saeki N, Kuwahara Y, Sasaki H, Satoh H & Shiroishi T Gasdermin (Gsdm) Localizing to mouse chromosome 11 is predominantly expressed in upper gastrointestinal tract but significantly suppressed in human gastric cancer cells. Mamm. Genome 11, 718–724 (2000). - PubMed
-
- Delmaghani S et al. Mutations in the gene encoding pejvakin, a newly identified protein of the afferent auditory pathway, cause DFNB59 auditory neuropathy. Nat. Genet 38, 770–778 (2006). - PubMed
-
- Yu J et al. Polymorphisms in GSDMA and GSDMB are associated with asthma susceptibility, atopy and BHR. Pediatr. Pulmonol 46, 701–708 (2011). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

