Sound garden: How snakes respond to airborne and groundborne sounds
- PMID: 36787306
- PMCID: PMC9928108
- DOI: 10.1371/journal.pone.0281285
Sound garden: How snakes respond to airborne and groundborne sounds
Erratum in
-
Correction: Sound garden: How snakes respond to airborne and groundborne sounds.PLoS One. 2025 Apr 1;20(4):e0321508. doi: 10.1371/journal.pone.0321508. eCollection 2025. PLoS One. 2025. PMID: 40168264 Free PMC article.
Abstract
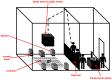
Evidence suggests that snakes can hear, but how snakes naturally respond to sound is still unclear. We conducted 304 controlled experiment trials on 19 snakes across five genera in a sound-proof room (4.9 x 4.9 m) at 27ºC, observing the effects of three sounds on individual snake behavior, compared to controls. We quantified eight snake behaviors (body movement, body freezing, head-flicks, tongue-flicks, hissing, periscoping, head fixation, lower jaw drop) in response to three sounds, which were filtered pink-noise within the following frequency ranges: 0-150Hz (sound 1, which produced ground vibrations, as measured by an accelerometer), 150-300Hz (sound 2, which did not produced ground vibrations), 300-450Hz (sound 3, which did not produced ground vibrations). All snake responses were strongly genus dependent. Only one genus (Aspidites, Woma Pythons) significantly increased their probability of movement in response to sound, but three other genera (Acanthophis (Death Adders), Oxyuranus (Taipans), and Pseudonaja (Brown Snakes)) were more likely to move away from sound, signaling potential avoidance behavior. Taipans significantly increased their likelihood of displaying defensive and cautious behaviors in response to sound, but three of the five genera exhibited significantly different types of behaviors in sound trials compared to the control. Our results highlight potential heritable behavioral responses of snakes to sound, clustered within genera. Our study illustrates the behavioral variability among different snake genera, and across sound frequencies, which contributes to our limited understanding of hearing and behavior in snakes.
Copyright: © 2023 Zdenek et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
-
- Young BA. A review of sound production and hearing in snakes, with a discussion of intraspecific acoustic communication in snakes. J Pa Acad Sci. 1997;71: 39–46.
-
- Wever EG, Vernon JA. The problem of hearing in snakes. J ofAuditory Res. 1960;1: 77–83.
MeSH terms
LinkOut - more resources
Full Text Sources

