New Approach Methodologies for the Endocrine Activity Toolbox: Environmental Assessment for Fish and Amphibians
- PMID: 36789969
- PMCID: PMC10258674
- DOI: 10.1002/etc.5584
New Approach Methodologies for the Endocrine Activity Toolbox: Environmental Assessment for Fish and Amphibians
Abstract
Multiple in vivo test guidelines focusing on the estrogen, androgen, thyroid, and steroidogenesis pathways have been developed and validated for mammals, amphibians, or fish. However, these tests are resource-intensive and often use a large number of laboratory animals. Developing alternatives for in vivo tests is consistent with the replacement, reduction, and refinement principles for animal welfare considerations, which are supported by increasing mandates to move toward an "animal-free" testing paradigm worldwide. New approach methodologies (NAMs) hold great promise to identify molecular, cellular, and tissue changes that can be used to predict effects reliably and more efficiently at the individual level (and potentially on populations) while reducing the number of animals used in (eco)toxicological testing for endocrine disruption. In a collaborative effort, experts from government, academia, and industry met in 2020 to discuss the current challenges of testing for endocrine activity assessment for fish and amphibians. Continuing this cross-sector initiative, our review focuses on the current state of the science regarding the use of NAMs to identify chemical-induced endocrine effects. The present study highlights the challenges of using NAMs for safety assessment and what work is needed to reduce their uncertainties and increase their acceptance in regulatory processes. We have reviewed the current NAMs available for endocrine activity assessment including in silico, in vitro, and eleutheroembryo models. New approach methodologies can be integrated as part of a weight-of-evidence approach for hazard or risk assessment using the adverse outcome pathway framework. The development and utilization of NAMs not only allows for replacement, reduction, and refinement of animal testing but can also provide robust and fit-for-purpose methods to identify chemicals acting via endocrine mechanisms. Environ Toxicol Chem 2023;42:757-777. © 2023 The Authors. Environmental Toxicology and Chemistry published by Wiley Periodicals LLC on behalf of SETAC.
Keywords: Adverse outcome pathway; Endocrine-disrupting compounds; In vitro toxicology.
© 2023 The Authors. Environmental Toxicology and Chemistry published by Wiley Periodicals LLC on behalf of SETAC.
Conflict of interest statement
Figures
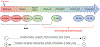


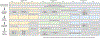
References
-
- Alcaraz AJG, Mikulášek K, Potěšil D, Park B, Shekh K, Ewald J, Burbridge C, Zdráhal Z, Schneider D, Xia J, Crump D, Basu N, & Hecker M (2021). Assessing the toxicity of 17α-ethinylestradiol in rainbow trout using a 4-day transcriptomics benchmark dose (BMD) embryo assay. Environmental Science & Technology, 55(15), 10608–10618. 10.1021/acs.est.1c02401 - DOI - PubMed
-
- Ankley GT, Bennett RS, Erickson RJ, Hoff DJ, Hornung MW, Johnson RD, Mount DR, Nichols JW, Russom CL, Schmieder PK, Serrrano JA, Tietge JE, & Villeneuve DL (2010). Adverse outcome pathways: A conceptual framework to support ecotoxicology research and risk assessment. Environmental Toxicology and Chemistry, 29(3), 730–741. 10.1002/etc.34 - DOI - PubMed
-
- Arena M, & van der Linden S (2021). Annex A—Use of the XETA in the assessment strategy of the ECHA/EFSA guidance.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

