Synaptotagmin-7 facilitates acetylcholine release in splanchnic nerve-chromaffin cell synapses during nerve activity
- PMID: 36796621
- PMCID: PMC10145958
- DOI: 10.1016/j.neulet.2023.137129
Synaptotagmin-7 facilitates acetylcholine release in splanchnic nerve-chromaffin cell synapses during nerve activity
Abstract
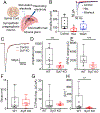
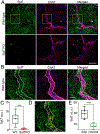
Disturbances that threaten homeostasis elicit activation of the sympathetic nervous system (SNS) and the adrenal medulla. The effectors discharge as a unit to drive global and immediate changes in whole-body physiology. Descending sympathetic information is conveyed to the adrenal medulla via preganglionic splanchnic fibers. These fibers pass into the gland and synapse onto chromaffin cells, which synthesize, store, and secrete catecholamines and vasoactive peptides. While the importance of the sympatho-adrenal branch of the autonomic nervous system has been appreciated for many decades, the mechanisms underlying transmission between presynaptic splanchnic neurons and postsynaptic chromaffin cells have remained obscure. In contrast to chromaffin cells, which have enjoyed sustained attention as a model system for exocytosis, even the Ca2+ sensors that are expressed within splanchnic terminals have not yet been identified. This study shows that a ubiquitous Ca2+-binding protein, synaptotagmin-7 (Syt7), is expressed within the fibers that innervate the adrenal medulla, and that its absence can alter synaptic transmission in the preganglionic terminals of chromaffin cells. The prevailing impact in synapses that lack Syt7 is a decrease in synaptic strength and neuronal short-term plasticity. Evoked excitatory postsynaptic currents (EPSCs) in Syt7 KO preganglionic terminals are smaller in amplitude than in wild-type synapses stimulated in an identical manner. Splanchnic inputs also display robust short-term presynaptic facilitation, which is compromised in the absence of Syt7. These data reveal, for the first time, a role for any synaptotagmin at the splanchnic-chromaffin cell synapse. They also suggest that Syt7 has actions at synaptic terminals that are conserved across central and peripheral branches of the nervous system.
Keywords: Adrenal medulla; Preganglionic synapses; Synaptotagmin; Synaptotagmin-7.
Copyright © 2023 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


Similar articles
-
PACAP regulates immediate catecholamine release from adrenal chromaffin cells in an activity-dependent manner through a protein kinase C-dependent pathway.J Neurochem. 2009 Aug;110(4):1214-25. doi: 10.1111/j.1471-4159.2009.06206.x. Epub 2009 Jun 5. J Neurochem. 2009. PMID: 19508428 Free PMC article.
-
Cholinergic and peptidergic neurotransmission in the adrenal medulla: A dynamic control of stimulus-secretion coupling.IUBMB Life. 2020 Apr;72(4):553-567. doi: 10.1002/iub.2117. Epub 2019 Jul 13. IUBMB Life. 2020. PMID: 31301221 Review.
-
Electrophysiological and morphological features underlying neurotransmission efficacy at the splanchnic nerve-chromaffin cell synapse of bovine adrenal medulla.Am J Physiol Cell Physiol. 2010 Feb;298(2):C397-405. doi: 10.1152/ajpcell.00440.2009. Epub 2009 Nov 25. Am J Physiol Cell Physiol. 2010. PMID: 19940070
-
Synaptotagmin 7 Mediates Both Facilitation and Asynchronous Release at Granule Cell Synapses.J Neurosci. 2018 Mar 28;38(13):3240-3251. doi: 10.1523/JNEUROSCI.3207-17.2018. J Neurosci. 2018. PMID: 29593071 Free PMC article.
-
A physiological view of the central and peripheral mechanisms that regulate the release of catecholamines at the adrenal medulla.Acta Physiol (Oxf). 2008 Feb;192(2):287-301. doi: 10.1111/j.1748-1716.2007.01807.x. Epub 2007 Nov 15. Acta Physiol (Oxf). 2008. PMID: 18005392 Review.
Cited by
-
Synaptotagmin-7 Counteracts Short-Term Depression during Phasic Dopamine Release.eNeuro. 2024 Mar 12;11(3):ENEURO.0501-23.2024. doi: 10.1523/ENEURO.0501-23.2024. Print 2024 Mar. eNeuro. 2024. PMID: 38365841 Free PMC article.
References
-
- Bader MF, Holz RW, Kumakura KONO, Vitale NICO, Exocytosis: The Chromaffin Cell As a Model System, Ann. N. Y. Acad. Sci 971 (1) (2002) 178–183. NOT IN FILE. - PubMed
-
- Bendahmane M, Morales A, Kreutzberger AJB, Schenk NA, Mohan R, Bakshi S, Philippe JM, Zhang S, Kiessling V, Tamm LK, Giovannucci DR, Jenkins PM, Anantharam A, Synaptotagmin-7 enhances calcium-sensing of chromaffin cell granules and slows discharge of granule cargos, J. Neurochem 154 (6) (2020) 598–617. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

