Youth with Cerebral Palsy Display Abnormal Somatosensory Cortical Activity During a Haptic Exploration Task
- PMID: 36796750
- PMCID: PMC10023489
- DOI: 10.1016/j.neuroscience.2023.01.030
Youth with Cerebral Palsy Display Abnormal Somatosensory Cortical Activity During a Haptic Exploration Task
Abstract
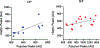
There are numerous clinical reports that youth with cerebral palsy (CP) have proprioceptive, stereognosis and tactile discrimination deficits. The growing consensus is that the altered perceptions in this population are attributable to aberrant somatosensory cortical activity seen during stimulus processing. It has been inferred from these results that youth with CP likely do not adequately process ongoing sensory feedback during motor performance. However, this conjecture has not been tested. Herein, we address this knowledge gap using magnetoencephalographic (MEG) brain imaging by applying electrical stimulation to the median nerve of youth with CP (N = 15, Age = 15.8 ± 0.83 yrs, Males = 12, MACS levels I-III) and neurotypical (NT) controls (N = 18, Age = 14.1 ± 2.4 yrs, Males = 9) while at rest (i.e., passive) and during a haptic exploration task. The results illustrated that the somatosensory cortical activity was reduced in the group with CP compared to controls during the passive and haptic conditions. Furthermore, the strength of the somatosensory cortical responses during the passive condition were positively associated with the strength of somatosensory cortical responses during the haptic condition (r = 0.75, P = 0.004). This indicates that the aberrant somatosensory cortical responses seen in youth with CP during rest are a good predictor of the extent of somatosensory cortical dysfunction during the performance of motor actions. These data provide novel evidence that aberrations in somatosensory cortical function in youth with CP likely contribute to the difficulties in sensorimotor integration and the ability to effectively plan and execute motor actions.
Keywords: MEG; hand; motor; sensorimotor integration; sensory feedback.
Copyright © 2023 IBRO. Published by Elsevier Ltd. All rights reserved.
Conflict of interest statement
Competing Interests
The authors declare no conflicts of interest.
Figures
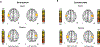
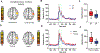
Similar articles
-
Aberrant movement-related somatosensory cortical activity mediates the extent of the mobility impairments in persons with cerebral palsy.J Physiol. 2022 Aug;600(15):3537-3548. doi: 10.1113/JP282898. Epub 2022 Jul 6. J Physiol. 2022. PMID: 35723200 Free PMC article.
-
The somatosensory cortical activity in individuals with cerebral palsy displays an aberrant developmental trajectory.J Physiol. 2021 Feb;599(4):1281-1289. doi: 10.1113/JP280400. Epub 2020 Dec 22. J Physiol. 2021. PMID: 33296078 Free PMC article.
-
Haptic exploration attenuates and alters somatosensory cortical oscillations.J Physiol. 2018 Oct;596(20):5051-5061. doi: 10.1113/JP276263. Epub 2018 Sep 21. J Physiol. 2018. PMID: 30152170 Free PMC article.
-
Somatosensory deficits and neural correlates in cerebral palsy: a scoping review.Dev Med Child Neurol. 2021 Dec;63(12):1382-1393. doi: 10.1111/dmcn.14963. Epub 2021 Jun 17. Dev Med Child Neurol. 2021. PMID: 34145582 Free PMC article.
-
Interrelationships of Touch and Proprioception with Motor Impairments in Individuals with Cerebral Palsy: A Systematic Review.Percept Mot Skills. 2022 Jun;129(3):570-590. doi: 10.1177/00315125221093904. Epub 2022 Apr 22. Percept Mot Skills. 2022. PMID: 35452588
Cited by
-
Contributions of the sensory system to motor learning : Focus on Cerebral Palsy.Pediatr Res. 2024 Jul 27. doi: 10.1038/s41390-024-03421-y. Online ahead of print. Pediatr Res. 2024. PMID: 39068271 No abstract available.
-
Somatosensory modulation of affective pictures' processing in adults with cerebral palsy and healthy controls: a case-control study.BMC Neurol. 2025 Feb 17;25(1):67. doi: 10.1186/s12883-025-04072-0. BMC Neurol. 2025. PMID: 39962417 Free PMC article.
-
Proprioception, Emotion and Social Responsiveness in Children with Developmental Disorders: An Exploratory Study in Autism Spectrum Disorder, Cerebral Palsy and Different Neurodevelopmental Situations.Children (Basel). 2024 Jun 13;11(6):719. doi: 10.3390/children11060719. Children (Basel). 2024. PMID: 38929298 Free PMC article.
References
-
- Angel RW and Malenka RC (1982). “Velocity-dependent suppression of cutaneous sensitivity during movement.” Exp Neurol 77(2): 266–274. - PubMed
-
- Auld ML, Boyd RN, Moseley GL, Ware RS and Johnston LM (2012). “Impact of tactile dysfunction on upper-limb motor performance in children with unilateral cerebral palsy.” Arch Phys Med Rehabil 93(4): 696–702. - PubMed
-
- Christensen D, Van Naarden Braun K, Doernberg NS, Maenner MJ, Arneson CL, Durkin MS, Benedict RE, Kirby RS, Wingate MS, Fitzgerald R and Yeargin-Allsopp M (2014). “Prevalence of cerebral palsy, co-occurring autism spectrum disorders, and motor functioning - Autism and Developmental Disabilities Monitoring Network, USA, 2008.” Dev Med Child Neurol 56(1): 59–65. - PMC - PubMed
-
- Clayton K, Fleming JM and Copley J (2003). “Behavioral responses to tactile stimuli in children with cerebral palsy.” Phys Occup Ther Pediatr 23(1): 43–62. - PubMed
-
- Condliffe EG, Jeffery DT, Emery DJ, Treit S, Beaulieu C and Gorassini MA (2019). “Full Activation Profiles and Integrity of Corticospinal Pathways in Adults With Bilateral Spastic Cerebral Palsy.” Neurorehabil Neural Repair 33(1): 59–69. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

