This is a preprint.
A non-canonical striatopallidal "Go" pathway that supports motor control
- PMID: 36798372
- PMCID: PMC9934763
- DOI: 10.21203/rs.3.rs-2524816/v1
A non-canonical striatopallidal "Go" pathway that supports motor control
Update in
-
A non-canonical striatopallidal Go pathway that supports motor control.Nat Commun. 2023 Oct 23;14(1):6712. doi: 10.1038/s41467-023-42288-1. Nat Commun. 2023. PMID: 37872145 Free PMC article.
Abstract
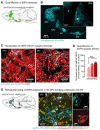
In the classical model of the basal ganglia, direct pathway striatal projection neurons (dSPNs) send projections to the substantia nigra (SNr) and entopeduncular nucleus to regulate motor function. Recent studies have re-established that dSPNs also possess "bridging" collaterals within the globus pallidus (GPe), yet the significance of these collaterals for behavior is unknown. Here we use in vivo optical and chemogenetic tools combined with deep learning approaches to dissect the roles of bridging collaterals in motor function. We find that dSPNs projecting to the SNr send synchronous motor-related information to the GPe via axon collaterals. Inhibition of native activity in dSPN GPe terminals impairs motor activity and function via regulation of pallidostriatal Npas1 neurons. We propose a model by which dSPN GPe collaterals ("striatopallidal Go pathway") act in concert with the canonical terminals in the SNr to support motor control by inhibiting Npas1 signals going back to the striatum.
Keywords: Go pathway; Npas1; arkypallidal; axon collaterals; axonal copy; basal ganglia; bridging collaterals; direct pathway; efference copy; globus pallidus; medium spiny neurons; motor control; striatopallidal; striatum.
Figures








References
-
- Klaus A., Alves da Silva J. & Costa R. M. What, If, and When to Move: Basal Ganglia Circuits and Self-Paced Action Initiation. Annu. Rev. Neurosci. 42, 459–483 (2019). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

