Multiancestry Genome-Wide Association Study of Aortic Stenosis Identifies Multiple Novel Loci in the Million Veteran Program
- PMID: 36802703
- PMCID: PMC10806851
- DOI: 10.1161/CIRCULATIONAHA.122.061451
Multiancestry Genome-Wide Association Study of Aortic Stenosis Identifies Multiple Novel Loci in the Million Veteran Program
Abstract
Background: Calcific aortic stenosis (CAS) is the most common valvular heart disease in older adults and has no effective preventive therapies. Genome-wide association studies (GWAS) can identify genes influencing disease and may help prioritize therapeutic targets for CAS.
Methods: We performed a GWAS and gene association study of 14 451 patients with CAS and 398 544 controls in the Million Veteran Program. Replication was performed in the Million Veteran Program, Penn Medicine Biobank, Mass General Brigham Biobank, BioVU, and BioMe, totaling 12 889 cases and 348 094 controls. Causal genes were prioritized from genome-wide significant variants using polygenic priority score gene localization, expression quantitative trait locus colocalization, and nearest gene methods. CAS genetic architecture was compared with that of atherosclerotic cardiovascular disease. Causal inference for cardiometabolic biomarkers in CAS was performed using Mendelian randomization and genome-wide significant loci were characterized further through phenome-wide association study.
Results: We identified 23 genome-wide significant lead variants in our GWAS representing 17 unique genomic regions. Of the 23 lead variants, 14 were significant in replication, representing 11 unique genomic regions. Five replicated genomic regions were previously known risk loci for CAS (PALMD, TEX41, IL6, LPA, FADS) and 6 were novel (CEP85L, FTO, SLMAP, CELSR2, MECOM, CDAN1). Two novel lead variants were associated in non-White individuals (P<0.05): rs12740374 (CELSR2) in Black and Hispanic individuals and rs1522387 (SLMAP) in Black individuals. Of the 14 replicated lead variants, only 2 (rs10455872 [LPA], rs12740374 [CELSR2]) were also significant in atherosclerotic cardiovascular disease GWAS. In Mendelian randomization, lipoprotein(a) and low-density lipoprotein cholesterol were both associated with CAS, but the association between low-density lipoprotein cholesterol and CAS was attenuated when adjusting for lipoprotein(a). Phenome-wide association study highlighted varying degrees of pleiotropy, including between CAS and obesity at the FTO locus. However, the FTO locus remained associated with CAS after adjusting for body mass index and maintained a significant independent effect on CAS in mediation analysis.
Conclusions: We performed a multiancestry GWAS in CAS and identified 6 novel genomic regions in the disease. Secondary analyses highlighted the roles of lipid metabolism, inflammation, cellular senescence, and adiposity in the pathobiology of CAS and clarified the shared and differential genetic architectures of CAS with atherosclerotic cardiovascular diseases.
Keywords: aortic valve stenosis; genome-wide association study; genomics; lipoprotein(a).
Conflict of interest statement
Conflict of Interest Disclosures
SMD receives research support from RenalytixAI and personal consulting fees from Calico Labs, outside the scope of the current research. PN reports personal consulting fees from Amgen, Apple, AstraZeneca, Blackstone Life Sciences, Foresite Labs, Genentech, Novartis, and TenSixteen Bio, investigator-initiated grants from Apple, AstraZeneca, and Boston Scientific, is a co-founder of TenSixteen Bio, equity in TenSixteen Bio, geneXwell, and Vertex, and spousal employment at Vertex, all unrelated to the present work. CJO is an employee of Novartis Institutes for Biomedical Research. LCW receives research support from IBM to the Broad Institute.
Figures
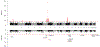

References
-
- Yadgir S, Johnson CO, Aboyans V, Adebayo OM, Adedoyin RA, Afarideh M, Alahdab F, Alashi A, Alipour V, Arabloo J, et al. Global, Regional, and National Burden of Calcific Aortic Valve and Degenerative Mitral Valve Diseases, 1990–2017. Circulation. 2020;141:1670–1680. doi: 10.1161/CIRCULATIONAHA.119.043391 - DOI - PubMed
-
- Dweck MR, Jones C, Joshi NV, Fletcher AM, Richardson H, White A, Marsden M, Pessotto R, Clark JC, Wallace WA, et al. Assessment of valvular calcification and inflammation by positron emission tomography in patients with aortic stenosis. Circulation. 2012;125:76–86. doi: 10.1161/CIRCULATIONAHA.111.051052 - DOI - PubMed
-
- Smith JG, Luk K, Schulz CA, Engert JC, Do R, Hindy G, Rukh G, Dufresne L, Almgren P, Owens DS, et al. Association of low-density lipoprotein cholesterol-related genetic variants with aortic valve calcium and incident aortic stenosis. JAMA. 2014;312:1764–1771. doi: 10.1001/jama.2014.13959 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
- S10 OD017985/OD/NIH HHS/United States
- UL1 TR000445/TR/NCATS NIH HHS/United States
- R01 HL139731/HL/NHLBI NIH HHS/United States
- I01 BX004821/BX/BLRD VA/United States
- R01 HL151283/HL/NHLBI NIH HHS/United States
- U01 HG011719/HG/NHGRI NIH HHS/United States
- S10 RR025141/RR/NCRR NIH HHS/United States
- R01 HL127564/HL/NHLBI NIH HHS/United States
- R01 HL148050/HL/NHLBI NIH HHS/United States
- R01 HL142711/HL/NHLBI NIH HHS/United States
- IK2 CX001780/CX/CSRD VA/United States
- R01 HL151152/HL/NHLBI NIH HHS/United States
- R01 HD074711/HD/NICHD NIH HHS/United States
- RC2 GM092618/GM/NIGMS NIH HHS/United States
- P50 GM115305/GM/NIGMS NIH HHS/United States
- U19 HL065962/HL/NHLBI NIH HHS/United States
- T32 HG010464/HG/NHGRI NIH HHS/United States
- S10 OD025092/OD/NIH HHS/United States
- U01 HG006378/HG/NHGRI NIH HHS/United States
- UL1 RR024975/RR/NCRR NIH HHS/United States
- R01 NS032830/NS/NINDS NIH HHS/United States
- U01 HG004798/HG/NHGRI NIH HHS/United States
- UL1 TR002243/TR/NCATS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Miscellaneous

