Inhaled prostacyclin therapy in the acute respiratory distress syndrome: a randomized controlled multicenter trial
- PMID: 36805707
- PMCID: PMC9938510
- DOI: 10.1186/s12931-023-02346-0
Inhaled prostacyclin therapy in the acute respiratory distress syndrome: a randomized controlled multicenter trial
Abstract
Background: Acute respiratory distress syndrome (ARDS) results in significant hypoxia, and ARDS is the central pathology of COVID-19. Inhaled prostacyclin has been proposed as a therapy for ARDS, but data regarding its role in this syndrome are unavailable. Therefore, we investigated whether inhaled prostacyclin would affect the oxygenation and survival of patients suffering from ARDS.
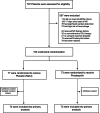
Methods: We performed a prospective randomized controlled single-blind multicenter trial across Germany. The trial was conducted from March 2019 with final follow-up on 12th of August 2021. Patients with moderate to severe ARDS were included and randomized to receive either inhaled prostacyclin (3 times/day for 5 days) or sodium chloride (Placebo). The primary outcome was the oxygenation index in the intervention and control groups on Day 5 of therapy. Secondary outcomes were mortality, secondary organ failure, disease severity and adverse events.
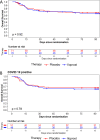
Results: Of 707 patients approached 150 patients were randomized to receive inhaled prostacyclin (n = 73) or sodium chloride (n = 77). Data from 144 patients were analyzed. The baseline PaO2/FiO2 ratio did not differ between groups. The primary analysis of the study was negative, and prostacyclin improved oxygenation by 20 mmHg more than Placebo (p = 0.17). Secondary analysis showed that the oxygenation was significantly improved in patients with ARDS who were COVID-19-positive (34 mmHg, p = 0.04). Mortality did not differ between groups. Secondary organ failure and adverse events were similar in the intervention and control groups.
Conclusions: The primary result of our study was negative. Our data suggest that inhaled prostacyclin might be beneficial treatment in patients with COVID-19 induced ARDS.
Trial registration: The study was approved by the Institutional Review Board of the Research Ethics Committee of the University of Tübingen (899/2018AMG1) and the corresponding ethical review boards of all participating centers. The trial was also approved by the Federal Institute for Drugs and Medical Devices (BfArM, EudraCT No. 2016003168-37) and registered at clinicaltrials.gov (NCT03111212) on April 6th 2017.
Keywords: ARDS; COVID-19; Inflammation; Prostacyclin; Therapy.
© 2023. The Author(s).
Conflict of interest statement
All authors declare that they do not have no conflicts of interest regarding this study.
Figures
References
-
- Ranieri VM, Pettila V, Karvonen MK, Jalkanen J, Nightingale P, Brealey D, Mancebo J, Ferrer R, Mercat A, Patroniti N, et al. Effect of intravenous interferon beta-1a on death and days free from mechanical ventilation among patients with moderate to severe acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2020;323(8):725–733. doi: 10.1001/jama.2019.22525. - DOI - PMC - PubMed
-
- Kor DJ, Carter RE, Park PK, Festic E, Banner-Goodspeed VM, Hinds R, Talmor D, Gajic O, Ware LB, Gong MN, et al. Effect of aspirin on development of ards in at-risk patients presenting to the emergency department: the LIPS—a randomized clinical trial. JAMA. 2016;315(22):2406–2414. doi: 10.1001/jama.2016.6330. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous